Innovative Solutions in Medication Packaging Can Improve Adherence
Drug Topics
SEPTEMBER 29, 2023
Near-field communication (NFC)-enabled medication blister packaging can help patients improve their medication adherence.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Drug Topics
SEPTEMBER 29, 2023
Near-field communication (NFC)-enabled medication blister packaging can help patients improve their medication adherence.
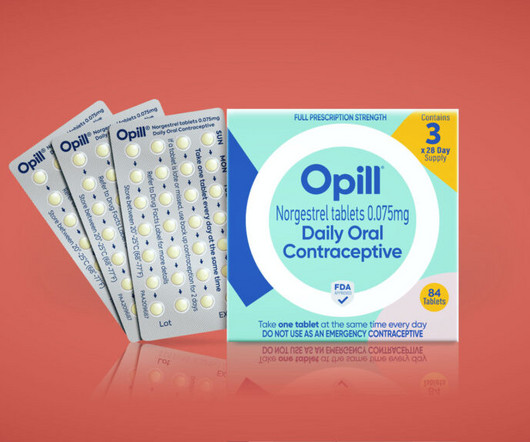
STAT
JULY 27, 2023
It’s not every day that a marketing team is tasked with designing the branding and packaging of the first over-the-counter birth control pill to be sold in the U.S.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Pharma Marketing Network
AUGUST 9, 2023
Navigating these regulatory challenges is essential to ensure compliance, maintain trust, and effectively communicate the benefits and risks of pharmaceutical products. Develop Clear and Balanced Messaging Effective communication is at the heart of pharmaceutical marketing. Keep thorough records of approvals for future reference.

European Pharmaceutical Review
OCTOBER 17, 2023
Assessing drug manufacturing facilities using alternative tools The FDA stated in the guidance that all manufacturing, packaging, and control sites for drug substance and drug product facilities are expected to be ready for inspection at the time of application submission.

pharmaphorum
OCTOBER 13, 2021
We are offering a generous salary plus a generous commission and bonus package, 25 days holiday and company pension scheme. Strong written and verbal communication skills. This role will give you an excellent opportunity to further develop and sharpen your commercial skills at all angles in a company that is currently expanding.

Pharma Marketing Network
AUGUST 9, 2023
Navigating these regulatory challenges is essential to ensure compliance, maintain trust, and effectively communicate the benefits and risks of pharmaceutical products. Develop Clear and Balanced Messaging Effective communication is at the heart of pharmaceutical marketing. Keep thorough records of approvals for future reference.

Pharma Mirror
AUGUST 8, 2020
Okapi Pharmacy, a wholly-owned subsidiary of Toppan Printing (Toppan) (TSE:7911), a global leader in communication, security, packaging, décor materials, and electronics solutions, has today announced the launch of Todokusuri EXPRESS, a service for delivering prescription medicines to patients through Todokusuri Yakkyoku, a pharmacy opened on July (..)

Pharmaceutical Technology
JUNE 19, 2023
It is critical to understand the supply chain, plan your packaging, storage and distribution lanes, then test the entire process before patient shipments go live. Products are stored and packaged in a dedicated suite by a dedicated team,” explains MacNeir. The requirements When handling personalised medicines, time is of the essence.

Pharmafile
MARCH 15, 2023
Some experts have explained that certain people could experience allergic reactions to pholcodine, and are recommending that the public check the packaging of any and all cough tablets or syrups they may have at home to check if pholcodine is within the ingredients. read more

DiversifyRx
NOVEMBER 24, 2023
This tracking involves the use of unique product identifiers, such as serial numbers and barcodes, on drug packaging to track their movement. Serialization Serialization is the process of assigning a unique identifier, typically a serial number, to each prescription drug package. The DSCSA aims to streamline this recall process.

pharmaphorum
OCTOBER 27, 2022
The overarching principle set out in Codes of Practice, and in particular the Principles for the use of digital channels in the EFPIA Code , is that the legislation and Codes of Practice apply equally to communications by companies on social media and digital channels.
European Pharmaceutical Review
DECEMBER 15, 2022
Communication with WHO encourage implementation of and accountability for the commitments and contributions toward improving access to insulin. Mikkelsen added that “the hope is to have insulin and diabetes devices as part of Essential Benefit Packages in low- and middle-income countries towards achieving Universal Health Coverage.”

The FDA Law Blog
MARCH 21, 2024
As the new law unfolds—and as state laws regarding ingredients and packaging, as well as laws regarding environmental claims continue to affect the cosmetics and personal care industries—the topics covered at the conference have never been more relevant.
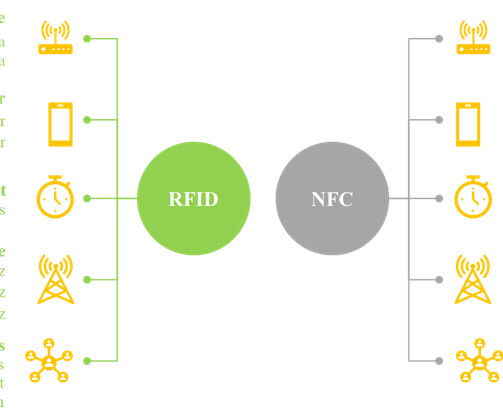
Roots Analysis
MARCH 8, 2023
In recent years, the use of smart labels allows the developer to convey a greater amount of information about the product to the consumers, without the need for additional packaging space. While most smartphones can read NFC chips, RFID tags can only be read by specialized receivers.

pharmaphorum
NOVEMBER 27, 2022
Supply chain challenges take many forms in pharma – from capacity issues, to managing excess safety stock, and ensuring accurate supplier communication. Better communication with traceable data. Automated systems allow suppliers to maintain complete traceability over their processes and communicate in the most effective way possible.

The FDA Law Blog
MARCH 8, 2023
For any major changes, additional time will be granted between the release of the implementation package and production deployment. To stay up to date with changes or review ones that have been implemented, refer to the regular communications, which can be found here.

The FDA Law Blog
JULY 10, 2023
FDA communicates via a Substantive Interaction to inform the submitter either that FDA will proceed with Interactive Review or that the 510(k) will be placed on hold until FDA receives a complete response to an Additional Information request. By Day 15 FDA conducts Acceptance Review. By Day 60 FDA conducts Substantive Review.

Pharmaceutical Technology
AUGUST 10, 2022
Expectations should be clearly communicated to the service providers as early as possible. Packaging needs?. Important items to look out for in the proposal include information on what was requested in terms of batch size, number of batches expected per year, component confirmation, and the packaging plan.???.

ISPE
MARCH 28, 2023
Thanks to the clinical supply chain service companies' years of industry development and operation, there are new developments in global supply chain layout, ultra-low temperature storage and distribution management breakthroughs, and growing packaging and labeling capabilities. By Li Jie of ClinsChain

ISPE
MARCH 28, 2023
Thanks to the clinical supply chain service companies' years of industry development and operation, there are new developments in global supply chain layout, ultra-low temperature storage and distribution management breakthroughs, and growing packaging and labeling capabilities. By Li Jie of ClinsChain

pharmaphorum
MAY 27, 2022
Teams and Zoom became the backbone of communication and firms embraced new ways of working. As a Non-Clinical Assessor at MHRA he assessed non-clinical data packages for new and generic medicines. About the interviewees.
pharmaphorum
JULY 22, 2021
We found that strong and open channels of communication were imperative. The company remained in close and frequent communication with regulatory agencies to accelerate procedures for the evaluation and authorisation of the clinical trials. We found that strong and open channels of communication were imperative” .

Aureus Medical Group
NOVEMBER 7, 2022
Just like no two travelers are alike, no two recruiters are the same, and you may need to work with a few before you find one with the communication, style, personality, and work ethic that meshes best with yours. Communicate quickly and clearly. . Answer: Yes, it’s perfectly okay to work with multiple recruiters!

DiversifyRx
NOVEMBER 30, 2023
Member-only perks such as free delivery or Rx packaging. Communication and Engagement Maintaining open lines of communication with members is essential. Effective communication ensures that members stay informed about the benefits of their membership and feel valued by your pharmacy.

Viseven
JUNE 12, 2023
Though South Korea is the third largest biopharma innovation hub, Suyoung admits that local pharma companies have low HCP engagement rates because marketers still use traditional methods of communication such as email, phone calls, and face-to-face visits.

Pharma Marketing Network
MARCH 25, 2023
All communications, both internal and external, should reflect the brand’s values and personality. This includes everything from the company’s website and social media presence to packaging and advertising. This can include creating a brand mission statement, defining brand values, and developing a brand personality.
European Pharmaceutical Review
OCTOBER 5, 2022
For most consumers, product packaging represents their first impression of a brand and its sustainability credentials. But how is this push shaping the pharma packaging market and what are the key changes we can expect to see? For many, aesthetic appeal and user experience top the list of priorities when it comes to packaging design.

pharmaphorum
SEPTEMBER 22, 2020
The pandemic has highlighted the difficulties in communicating with patients. A quick way to improve this would be to move to ‘digital by default’ communication”. The pandemic has highlighted the… difficulties in communicating with patients,” one respondent said. “A

European Pharmaceutical Review
JANUARY 25, 2024
7 Access to digital communications also remains a challenge in some Member States especially for those groups at risk of poverty and social exclusion (7.6 Even in countries with high smartphone usage such as Denmark, up to 25 percent of the population do not possess a smartphone.

European Pharmaceutical Review
MARCH 28, 2024
How does the qualification package support more meaningful endpoints and rare disease research as a result? Because the submission of the plan will open this dialogue, it will also socialise these issues with other rare disease populations and provide the opportunity to mitigate them in their own communities.

NF2 BioSolutions
JUNE 21, 2023
There was a small mix up with communication that Oscar had bloods done at school, so we had a couple of messages saying they were doing it at home, when in fact they were doing it at school as usual during term time. But we like these and will continue to use them when needed (ie not when its 27 degrees Celsius outside!).

The Checkup by Singlecare
MARCH 16, 2023
This includes counting, measuring, and packaging medications, as well as labeling and organizing them for distribution. They work closely with pharmacists to ensure that medications are prepared and dispensed accurately and safely,” he says.

DiversifyRx
NOVEMBER 3, 2022
All our cleared products have a package insert listed under the product, and all the information you need for the form is found there. In addition to selling tests, Kavel has solutions for online appointments, websites, and test results communication to patients or government entities. To make it easy, you can go to dbdiagnostics.com.

Aureus Medical Group
OCTOBER 19, 2022
It’s important to always be open and communicative with your travel companions, so everyone is in-tune and on track together. Aureus Medical Group travelers are covered from day one with a comprehensive perks and benefits package that includes: Low-cost, day-one health, vision, and dental. Unlimited loyalty and referral bonuses.

DiversifyRx
MARCH 22, 2024
Are they seniors interested in compliance packaging? Targeted marketing is a highly effective tool in this endeavor, allowing for personalized communication and tactics that resonate with specific audiences. Or busy professionals who would love delivery service? Are they parents in need of pediatric care products?

Roots Analysis
FEBRUARY 27, 2023
These market developments have made modular facilities more attractive to drug manufacturers today, as these facilities can quickly switch between multiple drugs, enabling formulation and packaging in multiple formats, such as solid, liquid, semi-solid and parenteral dosage forms.

pharmaphorum
AUGUST 17, 2020
“A lot of printed materials have been migrated to digital so that patients can opt-in to receive digital communications or HCPs can receive digital product or disease state information in the form of rep-triggered emails and microsites. Joanna Carlish. The idea is about really tapping into where the customer is in their journey.

Pharmaceutical Technology
NOVEMBER 9, 2022
With blockchain, every player in complex supply chains – from manufacturers to packaging plants to distribution warehouses – can log their contribution in a “block”, boosting both transparency and speed across a drug’s sprawling lifespan. Pharma firms who fail to incorporate AI will be left behind – they need to adapt now.
pharmaphorum
APRIL 11, 2022
Data will always be central to everything we do, as it has the power to communicate the potential of a new treatment to change a person’s life. Effective data analysis can lead to accelerated access and improved outcomes for patients.

ISPE
OCTOBER 1, 2022
Module Type Package: Turning Visionary Concepts into Reality. Module Type Package: Turning Visionary Concepts into Reality. The Module Type Package (MTP) approach designates each module with a digital description, enabling flexible module connection and orchestration. Trudy Patterson. Sat, 10/01/2022 - 06:00. Sponsored Content.
The Checkup by Singlecare
JANUARY 26, 2023
Hold their medication bottle or package and show them the relevant label information as you explain it. Remember that prescription labels are the last line of communication from a healthcare professional before a patient takes their medication. When counseling patients, don’t just talk at them.

pharmaphorum
AUGUST 11, 2020
“We can use the NHS as an example – look at the multidisciplinary nature of surgical procedures, for instance; the way they have to work, the way they communicate, the way they get feedback, the way they audit their results. That’s what we want to inject into Novo Nordisk.”.

The FDA Law Blog
NOVEMBER 2, 2023
The guidance describes elements that would be required of VCS bodies for recognition of standards they adopt: openness (with meaningful opportunities to participate), balance (broad range of stakeholders), due process, an appeals process, and consensus.

Aureus Medical Group
JULY 18, 2022
As you start to explore the process of traveling with Aureus Medical, it’s important to always communicate your anticipated travel timeline with your recruiter. Seasoned Aureus Medical Group travelers have applied for an assignment, interviewed, and set up housing arrangements in as little as one day! Are you on a strict, two-week timeframe?
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content