Cybin bags breakthrough tag for psychedelic depression drug
pharmaphorum
MARCH 14, 2024
Cybin files a $150m placement on the back of new clinical data with its psychedelic drug CYB003 for depression and an FDA breakthrough designation.

 tag depression
tag depression 
pharmaphorum
MARCH 14, 2024
Cybin files a $150m placement on the back of new clinical data with its psychedelic drug CYB003 for depression and an FDA breakthrough designation.

PharmaShots
JUNE 9, 2023
Quell Therapeutics Signed an Exclusive Option and License Agreement with AstraZeneca to Develop, Manufacture and Commercialize Engineered Treg Cell Therapies Date: June 09, 2023 | Tags: Quell Therapeutics, AstraZeneca, Engineered Treg Cell Therapies, Type 1 Diabetes, Inflammatory Bowel Disease, Biotech, Treg cell engineering modules Astellas and the (..)
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

PharmaShots
JANUARY 27, 2023
Magenta Therapeutics Pauses the P-I/II Study in AML Patients Date: Jan 27, 2023 | Tags: Magenta Therapeutics, MGTA-117, AML, Clinical Trial, P-I/II Ipsen Receives CHMP Negative Opinion for Palovarotene to Treat Fibrodysplasia Ossificans Progressiva Date: Jan 27, 2023 | Tags: Ipsen, Palovarotene, Fibrodysplasia Ossificans Progressiva, Regulatory, CHMP, (..)

PharmaShots
APRIL 14, 2023
Ocugen Reports Preliminary Results from the P-I/II Trial of OCU400 for the Treatment of Retinitis Pigmentosa and Leber Congenital Amaurosis Date: Apr 14, 2023 | Tags: Ocugen, OCU400, Retinitis Pigmentosa, Leber Congenital Amaurosis, Clinical Trial, P-I/II Trial Candesant Biomedical Receives the US FDA Clearance of Brella SweatControl Patch for Primary (..)

pharmaphorum
APRIL 26, 2021
Technology firm Jolly Good and Teijin Pharma have begun a partnership to develop virtual reality digital therapeutics (VR DTx) for major depressive disorder. However Nokia famously ran into trouble when it tried to develop VR products for digital health and its OZO camera failed to catch on, mainly because of a high $60,000 price tag.

pharmaphorum
AUGUST 21, 2022
Shares in Axsome Therapeutics have rocketed on FDA approval of its depression therapy Auvelity (formerly AXS-05) – a year after its approval was held up by the regulator. The post Axsome bounces back as FDA clears depression drug appeared first on. Photo by Sydney Sims on Unsplash.
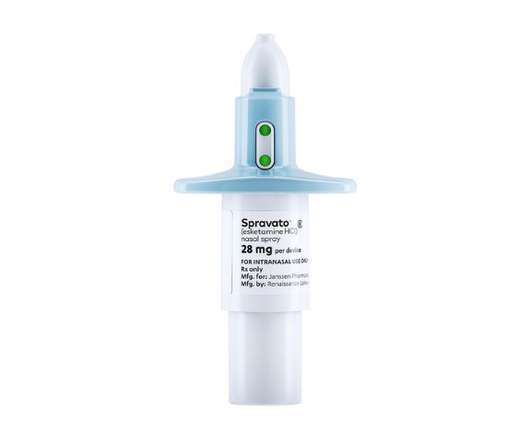
pharmaphorum
NOVEMBER 24, 2022
Johnson & Johnson’s Spravato therapy for treatment-resistant depression (TRD) hasn’t made the impact the company hoped for in the market, and it is looking to new clinical results to accelerate its take-up. Both drugs were given on top of standard antidepressants to patients wo had failed a least two earlier treatments.
Let's personalize your content