Walgreens issues statement on policy regarding abortion pill
Drug Store News
MARCH 7, 2023
Several politicians and the Biden administration have issued statements regarding Walgreens’ policy on the abortion pill.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 policy-issues
policy-issues 
Drug Store News
MARCH 7, 2023
Several politicians and the Biden administration have issued statements regarding Walgreens’ policy on the abortion pill.
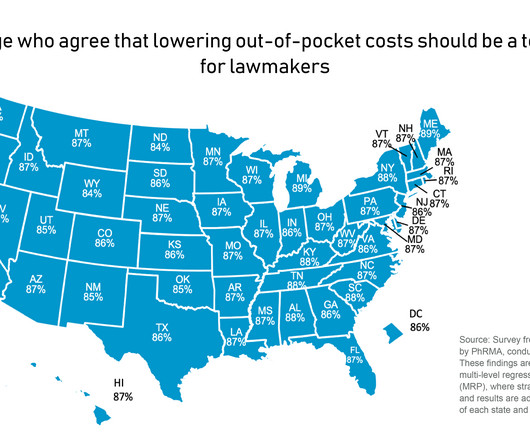
PhRMA
MAY 1, 2023
While policy debates about how to address these issues can seem never ending, American adults largely agree on what state policymakers should be doing to help deliver relief to patients at the pharmacy counter. Too many Americans are struggling to access and afford their health care.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

STAT
APRIL 18, 2024
Despite our different approaches, we believe that addressing health care costs is a truly bipartisan issue. One issue that is particularly ripe for bipartisan compromise is site-neutral payments. To be serious about creating access for people to the best possible care, that care must be affordable for patients and taxpayers.

Drug Topics
JANUARY 9, 2024
In August, the FDA issued a 1-year stabilization policy on the final enhanced drug distribution security requirements under the DSCSA. What exactly does that mean for pharmacists?

Fierce Healthcare
JUNE 14, 2023
The country’s largest physician organization is taking a strong stance against noncompete contracts for physicians and is redoubling efforts to urge Congress to boost Medicare payments. |

Pharmacy Times
JULY 27, 2022
You just need to tell your story and be genuine and be real, and it makes such a difference.

PhRMA
APRIL 14, 2023
As part of the implementation of the Inflation Reduction Act (IRA), the Centers for Medicare & Medicaid Services (CMS) issued draft guidance on the process for setting the price of medicines.

Fierce Pharma
AUGUST 16, 2023
Concern over the cost of prescription drugs is becoming less of a policy issue and more of a “core value” for voters across party lines. Concern over the cost of prescription drugs is becoming less of a policy issue and more of a “core value” for voters across party lines, a pollster found.
Drug Store News
NOVEMBER 30, 2023
The report summarizes the proceedings of CRN's annual Scientific Symposium, which brought together experts to shed light on the critical issues surrounding global nutrition challenges and policy efforts.

PhRMA
MARCH 1, 2023
Food and Drug Administration (FDA) are in the process of considering new collaboration initiatives aimed at biopharmaceutical patents.
STAT
APRIL 1, 2024
LONDON — Neurologists and patient advocates are up in arms over a policy decision by a U.K. And the issue is how the U.K.’s health agency that they say will imperil access to an ALS treatment that’s available in the U.S. and on its way to approval in the European Union.

Pharmacy Times
APRIL 19, 2023
The findings could inform revisions to safety guidelines and policies that help protect people with cardiovascular issues during heat waves.

Fierce Healthcare
JULY 17, 2023
Friday, the Federal Trade Commission (FTC) announced its withdrawal of two policy statements outlining its former position on antitrust enforcement in healthcare markets, echoing a similar decision | The two policy statements, issued in 1996 and 2011, "no longer reflect market realities in this important sector of the economy," the regulator (..)

STAT
SEPTEMBER 6, 2022
In a long-overdue move, the federal Office of Science and Technology Policy has issued guidance on making federally supported research and publications available to all without delay or embargo. These issues must be considered as this policy takes shape. Read the rest…

Drug Topics
FEBRUARY 2, 2023
Experts say Democrats and Republicans could work together on a variety of policy issues over the course of the year.

STAT
APRIL 4, 2024
government agency tasked with ensuring the safe use of radioactive materials issued a recent report that the agency’s advisers had key undisclosed conflicts of interest, raising fresh concerns about controversial decisions around the reporting of radioactive injection accidents. The oversight division of a U.S.

Pharmacy Times
SEPTEMBER 21, 2022
There are several important policy issues that could impact pharmacists’ ability to positively affect the quality of care for patients through the use of pharmacy performance measures.

STAT
OCTOBER 4, 2023
The committee has jurisdiction over Medicare and Medicaid payment policies for hospitals. Other committees that have tried tackling the issue have jurisdiction mostly over the Food and Drug Administration. The panel is tentatively aiming to hold a hearing on the issue in November, the sources said.

Hospital Pharmacy Europe
APRIL 4, 2024
The importance of pharmacy professionals signposting children and young people experiencing gender identity issues such as gender incongruence or dysphoria to appropriate support services has been emphasised by the General Pharmaceutical Council (GPhC).

STAT
NOVEMBER 9, 2023
Diagnosis, STAT’s twice-weekly newsletter about the politics and policy of health and medicine. And then there were five (well, six) A pared-back field of five GOP presidential candidates sparred Wednesday night, touching on health issues ranging from hot-button election topics, to wonky budget math.

STAT
JANUARY 25, 2024
Diagnosis, STAT’s twice-weekly newsletter about the politics and policy of health and medicine. They would be the first subpoenas issued by the Senate HELP committee in more than 40 years, if the vote happens and goes in Chair Bernie Sanders’ favor.

STAT
MARCH 7, 2024
Forty years of affirmative action policies were unable to compensate for the devastating impact of the 1910 Flexner Report, which led to the mass closure of medical schools that admitted Black students, and an estimated loss of 35,000 Black physicians into the field. But the truth is more complicated than that. Read the rest…

STAT
APRIL 18, 2024
The about-face illustrates the difficulty the biotech industry faces in dealing with an issue that, fueled by national security concerns, is moving fast and forcefully. It also tests the ability of Crowley, who lacks a background in politics, to navigate a domestic policy issue that is tangled up in U.S.-China China relations.
STAT
DECEMBER 11, 2023
The medical device lobby also announced the creation of a new division focused on policy issues for medical imaging technology. AdvaMed named Peter Arduini, CEO of GE HealthCare, chair of its board of directors on Monday. The announcement formalizes his position, which he will serve in for two years.

The FDA Law Blog
OCTOBER 5, 2023
Walsh — DOJ has issued another “voluntary disclosure” policy intended to encourage companies to disclose misconduct it discovers as part of a merger and acquisition. This new policy only applies to the disclosure of criminal conduct, not administrative or regulatory violations.

European Pharmaceutical Review
MARCH 12, 2024
Life science companies are investing in comprehensive data governance strategies to establish clear policies, procedures and controls for managing data. This involves regularly conducting internal audits and inspections to identify any potential gaps or non-compliance issues that need to be addressed. Training and education.

The FDA Law Blog
FEBRUARY 19, 2024
Loloei is a 14-year veteran of the FDA, where most recently she served as a Senior Regulatory Counsel in the Office of Policy at CDRH. Loloei also served as a Senior Policy Advisor in CDRH and as a Special Advisor in the Office of the Commissioner. (“HP&M”) is pleased to announce that Ana Loloei has joined the firm as Counsel.

STAT
JUNE 22, 2023
Doctors’ lobbyists in Washington tend to focus their energies on ensuring Medicare pays well, and on issues like so-called scope of practice laws. Whether from their lobbying or others, that first policy only forced researchers to make their work free after 12 months.

PhRMA
MAY 8, 2023
These policies work together to foster pediatric drug development, enabling biopharmaceutical companies to continue making significant investments in critical research areas for children. Food and Drug Administration (FDA) issued a BPCA written request to the sponsor for studies in pediatric cGVHD patients. In

STAT
AUGUST 1, 2023
Three large and growing physician groups backed by the private equity giant Welsh, Carson, Anderson & Stowe have hired a prominent lobbying firm to influence federal policy covering mergers and acquisitions. Over the past two weeks, United Musculoskeletal Partners, U.S. Anesthesia Partners, and U.S. Forbes Tate also did not respond.

STAT
JULY 12, 2023
LONDON — The European Union on Wednesday issued an antitrust fine of $475 million to the U.S. The commission called Illumina’s actions “an unprecedented and very serious infringement” of the EU’s policies, including the “cornerstone” that regulators review deals that can alter the market.

The FDA Law Blog
DECEMBER 15, 2022
By Riëtte van Laack — On December 7, 2022, FDA announced the availability of the final guidance regarding the enforcement policy for homeopathic drug products. FDA concluded that the CPG limited its ability to act against unsafe homeopathic drug products and decided to develop a risk-based enforcement policy.

The FDA Law Blog
APRIL 8, 2024
Claud — CDER’s Office of Pharmaceutical Quality (OPQ) issued its 2023 Annual Report last week, and it’s an upbeat assessment of the Office’s policy and outreach efforts. Again this year, the Annual Report that was released last week did not feature much in the way of specific quality issues. By John W.M.

STAT
NOVEMBER 7, 2023
The move comes two months after the FTC issued a policy statement noting some drug companies have failed to follow requirements for listing patents in the Food and Drug Administration’s Orange Book in hopes of thwarting competition from lower-cost generic medicines.

STAT
FEBRUARY 17, 2023
In December, he was named chairman of a new 12-person congressional commission tasked with issuing a set of policy recommendations for how the U.S. This year, Kelly will have a chance to advocate more formally. government can keep the country’s biotech industry competitive and capable of serving national security needs.
STAT
SEPTEMBER 1, 2022
An expert committee that advises the Centers for Disease Control and Prevention on vaccine policy voted on Thursday to recommend use of updated Covid-19 vaccines. With the positive votes from the Advisory Committee on Immunization Practices, the issue now goes to CDC Director Rochelle Walensky.

Hospital Pharmacy Europe
SEPTEMBER 26, 2023
Environmental sustainability within pharmacy is the subject of a new policy statement published by the International Pharmaceutical Federation (FIP) as it calls for action on climate change. C or 2°C above pre-industrial levels.

STAT
AUGUST 30, 2023
At issue is the cost of bedaquiline, which is the backbone of different combination therapies used to treat multi-drug-resistant tuberculosis. But advocacy groups continue to argue that Johnson & Johnson, which makes the pill, has stymied wider access.

STAT
JULY 27, 2023
To narrow the nation’s deeply entrenched health disparities, a permanent entity with regulatory powers should be created by the president to oversee health equity efforts across the entire federal government, says a report issued Thursday by the National Academies of Science, Engineering, and Medicine.

STAT
MARCH 14, 2023
At issue is the NRC’s policy that exempts practitioners of nuclear medicine — clinicians who use radioactive pharmaceuticals to diagnose or treat disease — from having to report when they mistakenly inject radiopharmaceuticals into a patient’s tissue rather than into a vein as intended.

STAT
JANUARY 6, 2023
In Nebraska, the state’s inspector general of corrections requested that the prison explain a policy, obtained by STAT, requiring that incarcerated people sign a consent form that misrepresents the benefits of available hepatitis C treatments. Lawmakers in other states are pledging broader probes into the issue, too.
STAT
APRIL 12, 2023
The White House on Wednesday issued an unprecedented warning about xylazine , the veterinary tranquilizer increasingly found in the illicit opioid supply. Beginning today, the substance will be classified as an “emerging drug threat” — the first designation of its kind.

STAT
SEPTEMBER 20, 2023
The policy would phase out patents and regulatory exclusivities and, in place of intellectual property protections, the U.S. A measure was passed directing the National Academies of Sciences, Engineering, and Medicine to study the idea and was added to the Pandemic and All-Hazards Preparedness Act. So, why prizes and not patents?

STAT
AUGUST 11, 2023
WASHINGTON — A White House task force created to work on drug shortage reforms has yet to contact the lawmakers who are writing legislation to stem shortages — and it’s not clear when the task force will recommend policies.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content