The Stories That Shine
PharmExec
SEPTEMBER 3, 2023
As someone who’s worked in pharma journalism for many years, there’s something extraordinary to hear about the why for individuals.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 journals pharmaceutical-executive
journals pharmaceutical-executive 
PharmExec
SEPTEMBER 3, 2023
As someone who’s worked in pharma journalism for many years, there’s something extraordinary to hear about the why for individuals.

STAT
OCTOBER 20, 2023
… China formally arrested an Astellas Pharma executive from Japan who had been detained since March , The Wall Street Journal writes. Hirokazu Matsuno, Japan’s chief cabinet secretary, said the executive, whom China previously identified as Hiroshi Nishiyama, was arrested in mid-October.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
STAT
FEBRUARY 9, 2023
… Pharmaceutical executives are ready to make deals , The Wall Street Journal says. Executives at Pfizer, Merck, and Novartis are looking for promising drugs to add to their pipelines and replenish sales as top-selling products lose patent protection in the coming years. To help you along, here are some tidbits.

European Pharmaceutical Review
NOVEMBER 27, 2023
NICE’s recommendation means that “babies born with Wolman Disease and their parents access to a new treatment option,” Bob Stevens, Group Chief Executive of the MPS Society commented. Evidence of successful long-term management of the disease was reported in a case study published last year in the Canadian Liver Journal.
STAT
JANUARY 9, 2023
Moreover, this is a particularly busy start to the week for those of you scrambling about at JPM, which is essentially a Woodstock for pharmaceutical executives and finance types. We hope the weekend respite was relaxing and invigorating, because that oh-so familiar routine of online meetings and deadlines has resumed.

European Pharmaceutical Review
NOVEMBER 28, 2023
The FDA’s approval of Ogsiveo is based on results from the Phase III DeFi clinical trial , which were published this year in the New England Journal of Medicine. The post First pill for desmoid tumours approved appeared first on European Pharmaceutical Review. Patients were randomised to receive 150mg of Ogsiveo or placebo. .

pharmaphorum
NOVEMBER 7, 2022
With more than a decade of experience leading and scaling managed care organisations, Grabski is a seasoned business leader and finance executive. He has also served as VP of Strategy and Execution, primarily responsible for adapting UHC’s Medicare Advantage business to the changes from the ACA. billion in revenue and 300 FTEs.
STAT
OCTOBER 20, 2022
The Wall Street Journal reports. Yet Yet the lobbying comes without some key lawmakers in the industry’s corner, and at a moment when one of its biggest trade groups, the Biotechnology Innovation Organization, must find a new leader after its chief executive clashed with board members and resigned. BIO

European Pharmaceutical Review
JULY 25, 2023
. “[It] is the first investigational chronic GVHD treatment to target inflammation and fibrosis through the inhibition of disease associated macrophages,” stated Michael A Metzger, Chief Executive Officer of Syndax Pharmaceuticals. Phase I/II data for axatilimab in GVHD has been published in the Journal of Clinical Oncology.

STAT
JUNE 30, 2023
Food and Drug Administration approved a gene therapy from BioMarin Pharmaceutical to treat people with hemophilia A, an inherited and rare bleeding disorder, STAT writes. The One of the pharmaceutical industry’s dealmakers is striking again to revive Bausch + Lomb, the eye-care company , The Wall Street Journal reports.
European Pharmaceutical Review
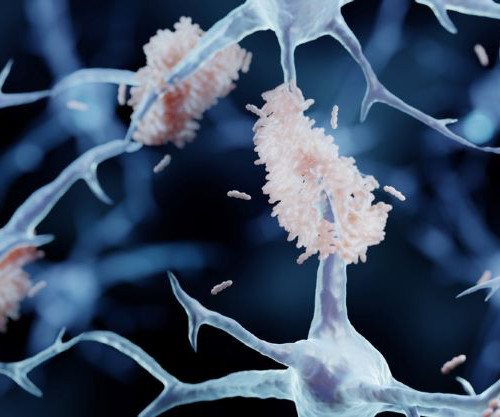
JANUARY 4, 2024
The post Novel drug delivery method shows promise in Alzheimer’s appeared first on European Pharmaceutical Review. Focused ultrasound therefore has “great promise for improving drug delivery to the brain” he added. Yet several barriers in the body must be overcome to reach its target site, such as the blood-brain barrier.

European Pharmaceutical Review
NOVEMBER 27, 2023
NICE’s recommendation means that “babies born with Wolman Disease and their parents access to a new treatment option,” Bob Stevens, Group Chief Executive of the MPS Society commented. Evidence of successful long-term management of the disease was reported in a case study published last year in the Canadian Liver Journal.

pharmaphorum
JULY 21, 2022
The pharmaceutical industry has long been highly regulated to help ensure quality and safety across all aspects of its business. Implementing Pharmaceutical Quality systems. If pharmaceutical companies have a fully operational and robust quality system, they will likely always be compliant with their region’s regulations.
European Pharmaceutical Review
MAY 24, 2023
Data from the Phase III (GEM-3) trial, published in the New England Journal of Medicine , demonstrated achievement of complete wound healing at six months and complete wound healing at three months. The post First redosable gene therapy approved appeared first on European Pharmaceutical Review. The “landmark approval” declared Krish S.

European Pharmaceutical Review
FEBRUARY 22, 2023
“Today’s approval offers physicians and patients in the EU more flexibility to determine the right treatment plan and enables more patients to potentially benefit from this medicine,” explained Dave Fredrickson, Executive Vice President of AstraZeneca’s Oncology Business Unit.

European Pharmaceutical Review
JANUARY 20, 2023
The safety profile of donanemab was initially reported from the TRAILBLAZER-ALZ trial in the New England Journal of Medicine. ” First virtual clinical trial examines two promising Alzheimer’s drugs The post FDA issues complete response letter for donanemab appeared first on European Pharmaceutical Review. .
European Pharmaceutical Review
JULY 17, 2023
The data was shared at the 2023 Alzheimer’s Association International Conference (AAIC) and published in the Journal of the American Medical Association (JAMA). The post New data suggests novel mAb could delay progression of early Alzheimer’s appeared first on European Pharmaceutical Review.
European Pharmaceutical Review
JANUARY 26, 2023
The trial results were presented at the American Society of Clinical Oncology 2022 Annual Meeting and published in The New England Journal of Medicine. The post Enhertu approved in EU for HER2-low breast cancer appeared first on European Pharmaceutical Review.
European Pharmaceutical Review
AUGUST 1, 2022
A study published in the Journal of the American Medical Association found that 12.4 The DDiCT initiative aligns with the pharmaceutical company’s broader diversity and inclusion goals for 2020-2025, which aim to address health disparities, clinical trial diversity, supplier diversity, employee giving and workforce representation. “All

The Honest Apothecary
MAY 3, 2023
. “Research published in the Journal of the American Medical Association (JAMA) Internal Medicine found that a chatbot’s responses to patient questions, pulled from a social media platform, were rated “significantly higher for both quality and empathy.” ’ MANAGED HEALTHCARE EXECUTIVE.
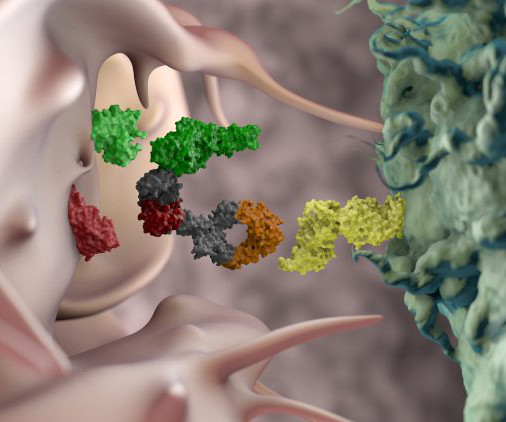
European Pharmaceutical Review
DECEMBER 12, 2023
Yannis Morel, Executive Vice President of product portfolio strategy and business development of Innate Pharma SA, offers insight on engineering antibodies to develop antibody-based NK cell therapeutics. Detailed results from COAST were published in the Journal of Clinical Oncology in 2022.

European Pharmaceutical Review
SEPTEMBER 29, 2022
The US Food and Drug Administration (FDA)’s Center for Drug Evaluation and Research (CDER) through the Office of Pharmaceutical Quality in 2021 embarked on an initiative to develop a framework for establishing a pharmaceutical manufacturing site quality rating system, the Quality management maturity (QMM) programme.

pharmaphorum
OCTOBER 19, 2022
He has held executive positions with several Fortune 500 companies and has been a featured thought leader in publications including CIO, WIRED, Forbes, Knowledge@Wharton, Los Angeles Times and The Wall Street Journal. Timothy White is the global head of digital at Teva Pharmaceuticals. Tim White, global head of digital, TEVA.

European Pharmaceutical Review
SEPTEMBER 26, 2022
Results published in The New England Journal of Medicine (NEJM) suggested marked improvement for patient quality of life, even in those with mild symptoms, hence the EC’s acceptance of the additional therapy. The trial evaluated the safety and efficacy of Ultomiris in 175 adult patients over a 26-week period.

pharmaphorum
MARCH 18, 2022
AstraZeneca (AZ), GlaxoSmithKline (GSK), Sanofi, Allergan and Britannia Pharma have been reprimanded for running advertisements that breached the Association of British Pharmaceutical Industry (APBI) code of practice. The post Latest ABPI code breaches reveal the risk of ‘likes’ on LinkedIn appeared first on.

PQA
JUNE 21, 2023
The guide was instrumental in identifying interventions and screenings that have addressed SDOH, specifically programs that are not described in peer-reviewed journals and other grey literature.

ISPE
JULY 6, 2023
In recent years, global pharmaceutical supply chains, still based on traditional batch- and intermediates-based manufacturing, were overwhelmed by their own vulnerabilities and inefficiencies, and were stressed to the brink of failure as a result of the SARS-COV-2 pandemic. In 2021, CONTINUUS Pharmaceuticals secured a$69.3

pharmaphorum
MAY 26, 2022
Ukrainian pharmaceutical companies are struggling to perform their duties in pharmacovigilance as Russia launched a wide-scale war in Ukraine. Russia’s full-scale invasion of Ukraine has already affected the global pharmaceutical industry a lot. This platform made work easier for many pharmaceutical companies.

PharmaShots
MARCH 9, 2023
Other Affiliations: Presently, Aida serves as an Executive Board Member for the International Science Reserve in The New York Academy of Sciences Board of Governors. Designation: Chief Executive Officer (CEO) and Chair of the Executive Board Previous company: Sanofi (8 yrs.) LinkedIn Total experience: 26 yrs.

pharmaphorum
JANUARY 27, 2022
The last chapter in Applied Virtual Reality in Healthcare, entitled “A Virtual Reality Platform for the Objective Measurement of Emotional State,” highlights potential obstacles ahead and examines how pharmaceutical companies can help address these issues. For example, mental health disorders and cognitive decline are rising.

pharmaphorum
OCTOBER 13, 2021
They will take responsibility for the accuracy and quality of the digital publication, direct the implementation of the editorial strategy and content plan by engaging with the in-house editorial team / freelancers / external partners to develop and execute high quality content for the website and its associated publications. Creative mind.

pharmaphorum
OCTOBER 13, 2021
The web editor is responsible for driving the editorial strategy and content plan (excluding the news channel) for pharmaphorum.com through engaging with the editorial team / freelancers / external partners to develop and execute high quality content for the website and its associated publications. BSc/BA in journalism or relevant field.

Viseven
OCTOBER 26, 2022
The pharmaceutical industry still faces significant challenges and is looking for innovative solutions. Below are five of the most significant pharmaceutical industry problems. . Therefore, to reduce the challenges of their pharmaceutical supply chain, they changed their strategy and made the most use of the resources available.

ISPE
SEPTEMBER 11, 2023
Herein, DM refers to a manufacturing model in which many units or facilities are disseminated across different geographical areas 6 , 7 DM facilities may produce similar or identical products under a unified pharmaceutical quality system (PQS) or operate as independent units with different outputs. Journal of Pharmaceutical Sciences 111, no.

pharmaphorum
DECEMBER 11, 2022
So, when a life sciences company embarks on a project of digital transformation or innovation, it’s really embarking on multiple projects that span the wide world of pharmaceutical operations. In 2019, he was awarded “Excellence in Medical Affairs Award” by the Association of Pharmaceutical Medicine Singapore (APMS).
IDStewardship
AUGUST 1, 2023
His main research interests are implementing clinical pharmacy/pharmaceutical care in infectious disease, intensive care and internal medicine. He has more than 30 articles published in SCI-e and ESCI indexed journals. He has been working as a committee member in the Antimicrobial Stewardship Team at the Hacettepe University Hospitals.

The People's Pharmacy
MAY 8, 2023
FDA Inspections Suspended: During the COVID pandemic, the Food and Drug Administration suspended its inspections at most foreign pharmaceutical facilities. ProPublica performs “investigative journalism in the public interest.” According to ProPublica, the FDA visited 45% of the pharmaceutical plants there in 2019.
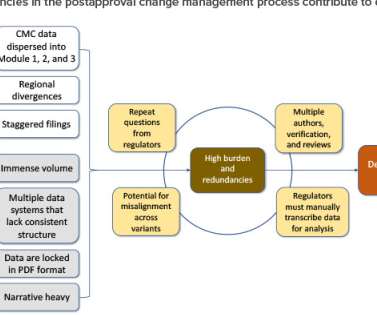
ISPE
AUGUST 29, 2022
Postapproval change management of pharmaceuticals is an essential part of life-cycle management but is associated with regulatory challenges. In an attempt to address these challenges, in 2019 the ICH endorsed ICH Q12, Technical and Regulatory Considerations for Pharmaceutical Product Lifecycle Management (and Annexes). Abernathy, J.

PharmaShots
MAY 29, 2023
The list of Highly Cited Researchers, especially in fields relevant to the pharmaceutical industry, can be an important source of intelligence on top talent that may be targets for recruitment or for research collaboration. David Pendlebury: There are several.
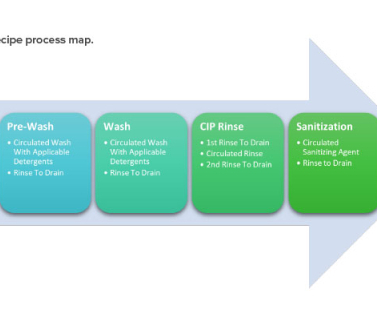
ISPE
JANUARY 11, 2023
5 It includes cleaning validation master plan, product risk assessment, utility and equipment readiness, analytical method readiness, sampling site selection/grouping, standard operating procedures, validation protocols, execution of validation protocols, personnel training, and the validation documentation package. 8 Hadziselimovic, D.,

Pharma Marketing Network
FEBRUARY 4, 2021
Desiree Priestley , Director, Patient Support Strategy & Insights at Otsuka America Pharmaceutical. HCPs still use and want journals in their offices, so it’s important to still consider what’s effective, even if it seems outdated. Ryan Billings , U.S. Oncology Digital Marketing & Customer Experience at GSK.

ISPE
JULY 7, 2023
4 , 5 Other challenges reflect the cautious nature of pharmaceutical manufacturing, like risk aversion and regulatory considerations. The organizing committee for the workshop included: Rich Chen, Executive Director, Purification & Viral Safety, Eli Lilly and Company Chris Hwang, Chief Technology Officer, Transcenta Therapeutics Inc.

European Pharmaceutical Review
JANUARY 25, 2023
The MAA from Vertex Pharmaceuticals and CRISPR Therapeutics is supported by two global Phase III studies. Earlier results from these ongoing trials were published in The New England Journal of Medicine in January 2021 and presented at the European Hematology Association Congress in June 2022.
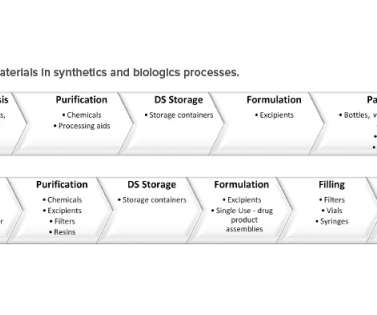
ISPE
AUGUST 30, 2022
The pharmaceutical manufacturing supply chain starts with the raw materials, which are needed to ensure drug availability for patients. The International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines contain information regarding regulatory requirements for raw materials.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content