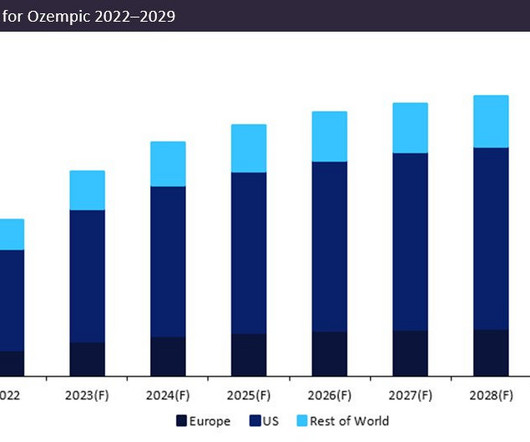
Injectable drug delivery market to value $1139.4b by 2029
European Pharmaceutical Review
MAY 7, 2024
percent compound annual growth rate (CAGR) between 2024 to 2029. billion by 2029. Approvals in the injectable drug delivery market In Nov 2023, the US Food and Drug Administration (FDA) approved the first and only obesity medication, Eli and Lilly’s Zepbound injection. by 2029 appeared first on European Pharmaceutical Review.























Let's personalize your content