GAO Releases Report on States’ Regulation of PBMs
Drug Topics
APRIL 19, 2024
Representatives from the US Government Accountability Office (GAO) interviewed regulators from 5 states on pharmacy benefit manager laws.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 law law-us
law law-us 
Drug Topics
APRIL 19, 2024
Representatives from the US Government Accountability Office (GAO) interviewed regulators from 5 states on pharmacy benefit manager laws.

Fierce Pharma
DECEMBER 15, 2022
Pfizer's Supreme Court challenge of US kickback law is 'far-fetched,' HHS says. Thu, 12/15/2022 - 14:50.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Drug Topics
JANUARY 24, 2024
As per US law Section 505(o)(4), the manufacturers are required to make the requested changes, respond with a modified version of the requested changes for review, or respond with a rebuttal within 30 days of the FDA's ask.

STAT
MAY 7, 2024
Please feel free to join us. appeals court must weigh how far FDA regulations for an abortion pill go in preempting state laws that regulate the practice of medicine — a largely untested area that legal analysts say will shape abortion access , Bloomberg Law explains. Best of luck, and do stay in touch.

Pharmacy Times
NOVEMBER 15, 2023
In this episode, you'll hear a conversation with Ron Lanton, partner at Lanton Law, who gets us up to speed on all of the legal cases surrounding this issue thus far.

STAT
NOVEMBER 13, 2023
Last week those of us who work in reproductive health care celebrated a historic victory in Ohio: Voters overwhelmingly favored Issue 1, which enshrines a right to abortion into the state constitution and makes the six-week ban many of us feared unenforceable. Read the rest…

STAT
JANUARY 26, 2024
Bernie Sanders jabbed Merck chief executive officer Robert Davis for telling congressional staff that he did not have the expertise to testify at a Senate hearing on prescription drug prices because he is a tax attorney , STAT tells us. The group argued the law runs afoul of the U.S. Continue to STAT+ to read the full story…

STAT
OCTOBER 31, 2023
Genetic testing companies and biotechnology groups are lobbying for revisions to a new Montana law that is more stringent than other state requirements intended to protect a consumer’s genetic data privacy, including additional oversight for increasingly popular at-home kit s, Bloomberg Law tells us.

The FDA Law Blog
NOVEMBER 15, 2023
(“HP&M”) is proud to announce the firm has been selected to the 2 024 edition of Best Law Firms. Firms included in the 2024 Best Law Firms ranking are recognized for professional excellence with persistently impressive ratings from clients and peers.

STAT
OCTOBER 11, 2022
I’m part of the sandwich generation: raising 9-year-old twin boys and helping take care of my 87-year-old mother-in-law, who lives with us. Before my father-in-law died, he had his wounds dressed right in my living room. Home health care workers make such care a reality for many families, including mine.

STAT
AUGUST 22, 2023
Below find a few items to get you started, and, as always, let us know if you hear anything interesting out there. The Food and Drug Administration approved a Pfizer vaccine that aims to protect newborns against RSV by vaccinating pregnant people, STAT tells us. Continue to STAT+ to read the full story…

STAT
SEPTEMBER 18, 2023
As you might expect, we are coping by quaffing a few cups of welcome stimulation — our flavor today is southern pecan — and, as always, we invite you to join us. The case is likely the first test of a barrage of lawsuits from the pharmaceutical industry and its allies over the new law. Chamber of Commerce and the U.S.

Pharmaceutical Technology
FEBRUARY 22, 2023
Amgen could be the winner of a high-stakes patent spat with Sanofi as tensions run high weeks before the March 27 US Supreme Court hearing. Since April 2020, Sanofi is responsible for Praluent outside of the US, while Regeneron handles its sale in the US. Amgen sued the two companies in 2014 over supposed patent infringement.

pharmaphorum
JANUARY 12, 2023
The Attorney General of Alabama has threatened to use an obscure law passed in 2006 to prosecute women who terminate a pregnancy using pharmacological means, shortly after the FDA made access to the drugs easier. ” The post Women in Alabama could face prosecution over use of abortion pills appeared first on.

STAT
AUGUST 30, 2023
While you ponder the possibilities, we invite you to join us for a delightful cup of stimulation. Please feel free to join us or, if you prefer, grab a bottle of water. So keep plugging away. After all, what are the alternatives? Our choice today is … cinnamon sticky bun. Have a wonderful day, and please do stay in touch.

pharmaphorum
JANUARY 20, 2022
US pharma giant Gilead Sciences has accused a network of unauthorised suppliers and distributors of selling more than $250 million of counterfeit versions of its HIV medications.

STAT
MAY 30, 2023
Please feel free to join us. Meanwhile, we hope you have a meaningful and productive day … Drugmakers have already hinted at suing Medicare over its new efforts to negotiate prescription drug prices, but a new report by nonpartisan congressional researchers suggests there are at least some parts of the law that cannot be challenged.

STAT
OCTOBER 25, 2023
So please join us as we hoist another cup of delicious stimulation. Have a grand day, and drop us a line if you hear something juicy. Ubl said future attacks are likely to be “applied challenges,” and over time, to target “the application” of the law, such as fines or other parts of the negotiation process.

The FDA Law Blog
NOVEMBER 6, 2023
That combined depth and breadth of experience is why clients come to us. The LMG Life Sciences awards are based on case evidence and feedback from clients and peers and selected by the editors to provide attorneys and law firms with information on the legal market and the U.S. life sciences industry.

The Guardian - Pharmaceutical Industry
MARCH 21, 2023
The three firms that control the insulin market are to cut costs in the US, but elsewhere users can spend all their pay to get the life-saving drug ‘I ration my insulin every month,” says Khushi Ahuja, a law student from Delhi who has type 1 diabetes and relies on human insulin manufactured by the US company Eli Lilly to manage her condition.

STAT
AUGUST 3, 2023
There is an opportunity to lead on this important issue as new tools enable us to transform aging. In 2011, President Obama signed the National Alzheimer’s Project Act into law, establishing a national goal of preventing and effectively treating the disease by 2025.

STAT
JUNE 22, 2023
” So while you tug on the ribbon, we will celebrate this notion by brewing still more cups of stimulation and invite you to join us. But an industry group expects a 25% decline in R&D and a drop in Europe’s global share of clinical trials from 25% to 19% if the law is passed. Our choice today is blueberry cobbler.

The FDA Law Blog
DECEMBER 21, 2023
This dinner—at the Waldorf Astoria in DC—will commemorate FDLI’s role during key eras in food and drug law and will have fun surprises as well!” May’s brother-in-law, songwriter Johnny Marks, later adapted the story into the famous song. Both are celebrating their 75th anniversaries in 2024. Mark your calendars now.

STAT
NOVEMBER 14, 2023
As for us, we are as busy as ever hunting and gathering items of interest. So join us as we hoist the ever-present cup of stimulation — our choice today is gingerbread — and attack the fast-growing to-do list. We trust you have your own hectic agendas. The U.S.

European Pharmaceutical Review
FEBRUARY 5, 2024
Incentivising small molecule drug innovation According to the Biotechnology Innovation Organization (BIO), the bipartisan Ensuring Pathways to Innovative Cures ( EPIC Act ) passed last week in the US, is a “critical” step for incentivising small molecule drug innovation.

The FDA Law Blog
MARCH 12, 2023
s panel of legal experts ( Karla Palmer , John Claud , Jeff Wasserstein , John Gilbert and Kalie Richardson ) invite you to spend your lunch hour (or hour-and-a-half…) with us as we address implications related to the looming end of the pandemic declaration. We hope you join us! Hyman, Phelps & McNamara, P.C.’s

STAT
AUGUST 16, 2023
The middle of the week is upon us. Securities and Exchange Commission examines whether Inotiv, a key importer, complied with a federal law that governs bribing foreign officials , STAT writes. The Have heart, though. You made it this far, so why not hang on for another couple of days, yes? A U.S.

STAT
MARCH 21, 2024
As always you are invited to join us. Right now, Medicare by law cannot cover obesity drugs, though it will pay for seniors to take the same medicines for conditions like diabetes and heart problems. For instance, we are scheduled to have a lively discussion about weight-loss drugs. Our choice today is orange creme.

pharmaphorum
JULY 22, 2022
The CMA has been looking into the activities of the two drugmakers for years, finding that they broke competition law in December 2016 and fining them £90 million – a verdict which was appealed by Pfizer and Flynn and was referred to the Competition Appeal Tribunal (CAT).

STAT
OCTOBER 2, 2023
As always, you are invited to join us. Meanwhile, Novo Nordisk became the latest pharmaceutical company to file a lawsuit against the Medicare drug price negotiation program , Bloomberg Law tells us. The Our choice is maple French toast. And of course, do keep in touch.

STAT
FEBRUARY 20, 2024
As always, you are invited to join us. Whatever your timetable, we hope you are feeling invigorated and inspired, because another busy day is on the way. To cope, yes, we are firing up the trusty coffee kettle and brewing cups of stimulation. Our choice today is toasted coconut mocha. We hope you have a smashing day and conquer the world.

pharmaphorum
FEBRUARY 18, 2022
The pandemic-induced transition to digital health is, according to the Nuffield Trust, contributing to an “inverse care law” that’s compounding existing health inequalities. Inverse care law. But, as demonstrated by initiatives like the Telehealth Equity Coalition (TEC), the sector is taking a united stand against inequitable access.

STAT
SEPTEMBER 20, 2023
The middle of the week is upon us. Meanwhile, do keep us in mind if you hear anything interesting. for selling unapproved versions of its Mounjaro diabetes drug, which is frequently used for combating obesity, STAT writes. Rise and shine, everyone. Have heart, though. And what better way to make the time fly than to keep busy.

STAT
OCTOBER 26, 2023
As always, you are invited to join us. We hope you find them useful and, more important, that you have a meaningful and productive day. … A broad push by federal, state, and private sectors to lower insulin costs in the U.S. There is an intensity to such mundane tasks, though, that calls for a cup of stimulation.

STAT
JULY 26, 2023
As for us, we are as busy as ever hunting and gathering items of interest. So join us as we hoist the ever-present cup of stimulation and attack the to-do list. We trust you have your own hectic agendas. Our choice today is cinnamon hazelnut.

STAT
JANUARY 22, 2024
As always, you are invited to join us. Many European countries have kept a restrictive stance when it comes to health system coverage of drugs that are primarily for weight loss, with some, including Germany, even ruling out such payments by law. The world, such as it is, continues to spin. Our choice today is peppermint mocha.

The FDA Law Blog
APRIL 21, 2024
Government—was that, despite having missed the statutory 60-day filing deadline, the patent was granted a PTE due to a change in the law created by Section 37 of the Leahy-Smith America Invents Act (“AIA”) ( Pub. which brings us to the topic of this post. Taiho Pharmaceutical Co.,
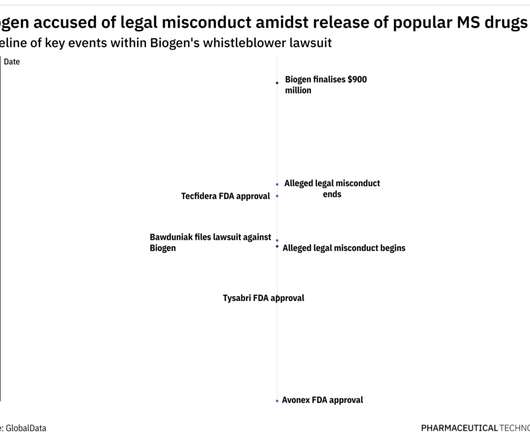
Pharmaceutical Technology
OCTOBER 5, 2022
As per the US Department of Justice, Biogen paid remuneration in the form of training and consulting fees and speaker honoraria, to induce physicians to prescribe the company’s drugs, in violation of the Anti-Kickback Statute. Under this law, relators such as Bawduniak can file lawsuits on behalf of the government.

STAT
SEPTEMBER 19, 2023
As for us, we are as busy as ever hunting and gathering items of interest. So join us as we hoist the ever-present cup of stimulation — our choice today is glazed doughnut — and attack the fast-growing to-do list. We trust you have your own hectic agendas.

Pharmaceutical Technology
JUNE 5, 2023
HIPAA laws are a series of federal regulatory standards relating to the Health Insurance Portability and Accountability Act of 1996, outlining the lawful use and disclosure of protected health information in the US. Pharmaceutical companies that are publicly traded will need to pay attention,” warns Kim.

pharmaphorum
SEPTEMBER 24, 2020
Gilead has paid $97 million to resolve claims by US authorities that it acted illegally by using a charitable foundation as a conduit to fund co-payments of thousands of Medicare patients taking its pulmonary arterial hypertension drug, Letairis. Gilead does not believe it violated the law and had no intention of doing so.

The FDA Law Blog
OCTOBER 4, 2023
Nevertheless, Judge Newman’s decision here gives us our first insights into the merits of the challenges against the Program. In contrast, Judge Newman found that Sixth Circuit (and Eighth Circuit) law established that “participation in Medicare, no matter how vital it may be to a business model, is a completely voluntary choice.”

STAT
DECEMBER 22, 2023
The end-of-year holiday break is upon us, so this, of course, is an opportunity to connect with special folks or, if need be, find some needed alone time. Drugmakers are slow-walking products to market to get around a federal law to lower medication prices, Bloomberg News explains. And what about you?

STAT
NOVEMBER 8, 2023
The middle of the week is upon us. Meanwhile, do keep us in mind if you hear anything interesting. The companies have 30 days to withdraw or amend their patent listings, or certify under penalty of perjury the listings comply with federal law. Rise and shine, everyone. Have heart, though.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content