From Point-in-time to Real-time
Pharmaceutical Commerce
JUNE 10, 2022
Why ‘real-time-all-the-time’ supply chain operations are fundamental to biopharma success.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 from-point-in-time-to-real-time
from-point-in-time-to-real-time 
Pharmaceutical Commerce
JUNE 10, 2022
Why ‘real-time-all-the-time’ supply chain operations are fundamental to biopharma success.
European Pharmaceutical Review
APRIL 9, 2024
In the investigation, a search query database was built to demonstrate “the deterministic nature of the bacterial strains with real and imaginary impedance, phase, and capacitance” It showed increased bacterial specification selectivity in the 9772.37 aeruginosa showed the greatest rise in real impedance and changes in phase A.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
ISPE
FEBRUARY 2, 2023
While RFID solutions have been in use for a long time, they still can fail to identify the object because of constraints of reading requirements of distance. Real Time Location Systems (RTLS) solution can work as a panacea to overcome those constraints. This will ensure an easier buy-in from the program sponsor.

pharmaphorum
JUNE 22, 2022
Pfizer has harnessed the expertise of health data specialist Truveta to provide close monitoring of safety and other signals for its products, including COVID-19 vaccine Comirnaty, almost in real-time. “We hope to continue to find additional benefit from this growing data such as identifying and addressing health disparities.”

IDStewardship
FEBRUARY 25, 2024
In this article four experts from bioMérieux discuss microbiology strategies to advance antimicrobial stewardship, reflecting on a recent Xchat (Twitter Chat) on the topic. Authored By: Katarzyna Wojcik, PMP, PhD, Andrea Prinzi, PhD, MPH, CPH, SM(ASCP), Amanda Suchanek, Ph.D., million deaths attributed to and 4.95

Fuld
DECEMBER 21, 2023
However, it’s estimated that over 50% of Battle Cards are out-of-date weakening their effectiveness in the face of real-time market dynamics. Risk of losing your competitive edge: Companies with old Battle Cards risk falling behind in a business environment where agility and timely insights are paramount.

European Pharmaceutical Review
JULY 13, 2023
The CDMO’s Carrie Mason shared that PAT, “takes a little bit more time and more effort and more capital to set up in the beginning, but once you get it up and running it’s very continuous and it should continue to provide results for a long time”. What is behind biopharma’s modern evolution?

European Pharmaceutical Review
APRIL 11, 2024
But as technology has advanced, we have arrived at a point where much of this work can be done virtually rather than with physical materials, saving time and material. Globally, the pharmaceutical sector emitted around 52 megatonnes of CO 2 in 2015, the most recent period for which detailed numbers are available.
European Pharmaceutical Review
JULY 20, 2023
An operation point with about 20 percent higher throughput was successfully operated at process variations. The authors stated their research highlighted the first time this kind of autonomous operation has been conducted. A model was developed from flow and coalescence models. How was the PAT study performed?

Digital Pharmacist
MARCH 13, 2023
Purchasing software can seem like a daunting task for pharmacy owners to tackle on top of all their other day-to-day tasks – but purchasing the right software can automate those tasks and thus free up so much valuable time in the long run. A pharmacy software program not only maximizes work efficiency but is also related to patient outcomes.
pharmaphorum
JANUARY 25, 2022
Oskar says OpenData provides their clients with customer reference data that helps interactions be more relevant and personalised with the right message, at the right time, on the right channel to meet HCPs unique needs. And limitations from some data providers on where and how companies can use data hinder rep productivity.

IDStewardship
JUNE 6, 2023
Algorithms tirelessly scouring vast databases to detect potential drug interactions, sparing us from accidental chemical calamities. We’ll dive into the many ways AI can assist pharmacists, from streamlining medication dispensing processes to enhancing patient education. Interview with: ChatGPT by OpenAI Interview by: Timothy P.
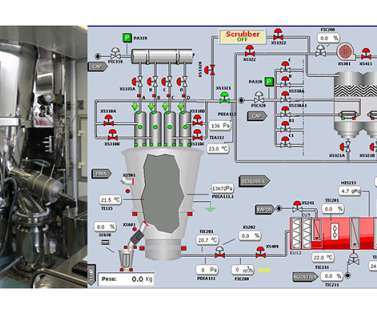
ISPE
NOVEMBER 17, 2022
The granules are dried by the combination of the diffusion of moisture from the solid, mediated by hot air, and the entrainment of this moisture by forced convection. The higher the temperature and the higher the intake air flow, the shorter the drying time. Artificial Intelligence Used to Optimize Fluid Bed Drying. Trudy Patterson.

Hospital Pharmacy Europe
MARCH 21, 2024
Ian Soulairol from the Institut Charles Gerhardt Montpellier at Montpellier University and pharmacist at the Department of Pharmacy of Nîmes University Hospital in France shares insights into how this technology is evolving and what it means for clinical practice now and in the future. These include printing time, temperature, etc.,

European Pharmaceutical Review
DECEMBER 6, 2023
He also shares some lessons from Dr Reddy’s efforts to increase automation of its manufacturing processes, and approaches for handing data. Elaborating on supply chain disruptions impacting sterile injectables, Venkatesh explains how the supply of e.g. packaging material and filters has not yet caught up demand following the pandemic.
Pharma Marketing Network
JULY 27, 2022
In the first two weeks after a cancer diagnosis, there are likely dozens of doctor’s visits, tests, consultations, and decision points. By helping them find relevant resources with highly specific information at the right time and in a chapterized format. Meeting Patients Where They Are. Supporting the Patient Journey.

pharmaphorum
DECEMBER 20, 2022
To make personalised healthcare a reality, data must be safely and responsibly used in a way that provides real value for patients and the entire healthcare ecosystem. To make personalised healthcare a reality, data must be safely and responsibly used in a way that provides real value for patients and the entire healthcare ecosystem.

ISPE
MARCH 30, 2023
However, for many organisations and facilities, being sold the advantages of digital twins, real-time predictive analytics, advanced robotics, and artificial intelligence, can feel like hearing a distant pipe dream. Greenfield facilities that have been the first to embrace the tools of Pharma 4.0 Industry 4.0,

The FDA Law Blog
DECEMBER 7, 2022
In simple terms from a non-scientist, these gene therapies add functional versions of genes to patients that do not have them, and thus the idea is to have these genes function in the same way as they do in individuals without these disorders. This product was developed by uniQure prior to being licensed to CSL Behring.

pharmaphorum
JULY 19, 2021
COVID has revolutionised the way pharma engages with healthcare stakeholders – but to be truly successful, digital engagement needs to move away from an overreliance on real-time meetings, say experts from Within3. This is despite in-person meetings having many obvious drawbacks.

European Pharmaceutical Review
FEBRUARY 8, 2024
A report from Prophecy Market Insights on rapid microbiology testing has predicted that the market is anticipated to surpass $12.7 Automation, data analytics, and real-time monitoring are some of the approaches supporting more innovative and efficient rapid microbiology testing methods, the authors remarked. billion by 2034.

pharmaphorum
OCTOBER 7, 2022
A UK study has found that Merck & Co’s oral COVID-19 therapy Lagevrio was unable to reduce hospitalisations compared to placebo in patients at higher risk from the virus, adding fuel to assertions in some quarters that its authorisation was premature. The European Commission has yet to approve it for use in the EU.

The Honest Apothecary
DECEMBER 16, 2023
“I want to commend each of you for being on time to this meeting today. Time is our most valuable asset, and when you show up on time it displays respect and appreciation for each other and for this organization.” But now even more so because I took the time to recognize that behavior.” “RPMs?”,

European Pharmaceutical Review
DECEMBER 14, 2023
Decisions to remove medicines from market are not taken lightly and are often based upon all evidence available to inform a benefit/risk balance. This evidence can come from a range of sources, such as randomised controlled trials (RCTs), observational studies, and spontaneous reporting. Meta-analyses contributed least frequently (31.4

OctariusRx
FEBRUARY 6, 2023
How to prevent drug diversion is something I spend significant time thinking about and talking about with my facilities. Whenever I have an opportunity to speak on this topic, I make it a point to remind everyone to expect diversion and develop systems before it happens. What are some of the common points of diversion?
pharmaphorum
AUGUST 3, 2022
We all know that real-world data (RWD) from electronic health records (EHR) could boost clinical trial efficiency and improve patient outcomes – so what’s holding us back? It’s been more than two decades since the ground-breaking potential of using electronic health records (EHR) to inform clinical research was first mooted.

ISPE
MARCH 6, 2023
Operations Data Challenges Limit the Value Realized from Pharma 4.0 Trudy Patterson Mon, 03/06/2023 - 11:50 iSpeak Blog iSpeak Operations Data Challenges Limit the Value Realized from Pharma 4.0 At the same time, many companies are pursuing localized manufacturing to adjust to supply chain challenges and to improve access to medicine.

Hospital Pharmacy Europe
JANUARY 11, 2024
In previous questionnaire-based analyses of clinicians responsible for fluid management – and with experience levels ranging from trainees to experienced clinicians – fluid management knowledge scores were low, and most participants reported having experienced unreported fluid-related serious adverse events.

IDStewardship
MARCH 23, 2023
In November 2022, #ASPchat partnered with bioMérieux and their experts, Patrick McDaneld, Brian Raux and Katherine Perez to address the gravity of this worldwide health concern in a real-time, open Twitter forum, highlighting microbiology labs and how they can impact antimicrobial stewardship program (ASP) successes.

pharmaphorum
JUNE 10, 2022
Pharma’s participation in end-to-end patient care through digital technologies can: Solve key treatment selection challenges to get the right patients prescribed the right therapies at the right time. Start thinking now about how to drive clinical impact, knowing that these clinical objectives will translate to increased revenue over time.

Omnicell
SEPTEMBER 5, 2022
Studies show that 1 in 10 healthcare workers will experience substance abuse at some point during their lives. Yet despite being the hardest hit by diversion, many health systems lack the necessary tools to detect these incidences in real-time. billion impact every year.

pharmaphorum
JANUARY 5, 2023
Touching upon telemedicine and learnings taken from the acute phase of the pandemic, Rossi discusses how systems can be far more flexible than was first thought. After all, cancer is not a disease that is going away – those patients are yet present and will be present within the health system.
pharmaphorum
JANUARY 11, 2023
However, the ‘Big Data paradigm’ is not so much concerned with the volume of that data, but how businesses and, indeed, industries can derive meaningful insights from what has become a glut of information. These training examples can be either real-world data or data synthetically generated by computer software,” he continued.
pharmaphorum
JULY 18, 2022
This means even more associated costs, resources, time, and effort. Import your organizational standards straight from your database into the clinical metadata repository. Already you’ve saved a huge amount of time getting started. Plus, now you won’t spend time searching all over for files and spreadsheets. No conflicts.

pharmaphorum
OCTOBER 18, 2022
The analysis showed that DCT studies enable time and cost efficiencies at virtually every point in the trial process. For many sponsors, understanding the tangible benefits of DCT studies is a great starting point. Integrating the patient journey. Failures in patient engagement and retention can delay or stop a clinical trial.

Viseven
DECEMBER 19, 2022
Pharma real-world evidence (RWE) has been in practice for decades; nevertheless, advances in digital technologies and analytics offer a new way to apply it. Also, it makes R&D (Research and Development) investments more efficient and accelerates time-to-market. What Is Real-World Evidence (RWE)?

ISPE
SEPTEMBER 10, 2023
Among global BoHs, variability in regulatory requirements and approval times for postapproval CMC changes has created an inordinately long delay between the first and last approval for a single CMC change for a pharmaceutical product. It also allows for comparisons between companies and across different periods of time. “In

pharmaphorum
DECEMBER 2, 2022
Using data-informed insights derived by effectively implemented datasets, artificial intelligence/machine learning capabilities, and technology sponsors can realise increased predictability in their study planning from protocol design through enrolment strategy. This emphasises sponsors’ need for enrolment predictability.

European Pharmaceutical Review
JANUARY 12, 2024
A recent survey from the Pistoia Alliance revealed that AI and machine learning (ML) will be the top technology investment for 60 percent of life sciences companies over the next two years. With proper oversight, GenAI has potential to drive efficiencies and cut the time needed for a regulatory submission, Levy says.

Pharmaceutical Technology
JUNE 15, 2023
Successful innovation requires enormous effort from countless project teams that build on the medical, scientific and technological research and development of generations before them. Product management AI in product management can improve every phase of a product, from ideation to development to commercialization.
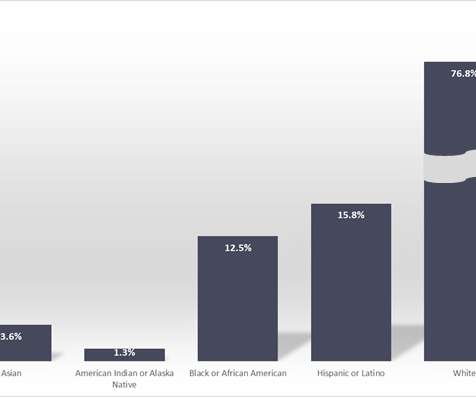
pharmaphorum
JUNE 27, 2022
The analysis – from a dataset that spans more than half a million patients – reveals once again the lack of inclusivity in clinical research, which undermines the ability of clinical trials to generate results that reflect the real-world situation in US cancer treatment.

Pharmaceutical Technology
OCTOBER 26, 2022
Several factors in recent years have contributed to this rise including more complex trial designs, the rapid adoption of decentralisation stemming from the pandemic, and larger global trials. Unplanned protocol deviations, which add time and cost to a study, are also contributors to complexity. Demand planning drives trial efficiency.

pharmaphorum
DECEMBER 29, 2022
I support the initiative of simplifying data privacy and the idea of making it more accessible, yet, there are some real challenges the Bill poses for organisations wishing to act from beyond the realms of the UK. This change could present a real risk to the free flow of personal data between the UK and the EU.
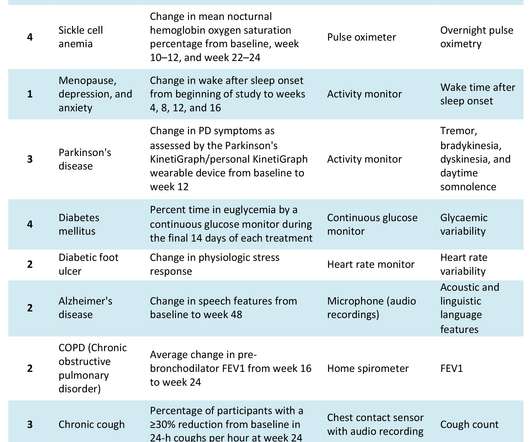
pharmaphorum
JULY 8, 2022
Preliminary research suggests that hybrid DCTs can reduce trial timing and resources , improve patient-centricity and patient engagement , and expand access and recruitment and increase diversity , mitigate bias, and reinforce inclusion in clinical trials. New sensor technology revolutionising data collection.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content