FDA Issues Two Guidance Documents on Food Allergen Labeling Requirements
The FDA Law Blog
DECEMBER 8, 2022
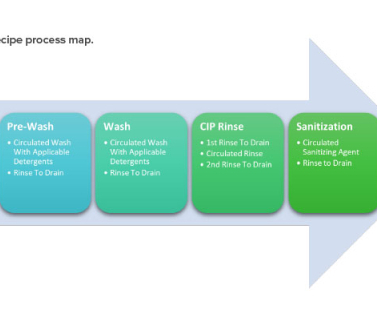
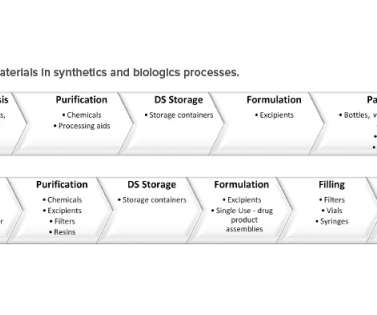
Food and Drug Administration (FDA) issued two guidance documents, one draft and one final, on food allergen labeling requirements. 1, 2023; The applicability of food allergen labeling requirements to specific products (e.g., 1, 2023; The applicability of food allergen labeling requirements to specific products (e.g.,
























Let's personalize your content