FDA warns patients about compounded versions of Novo Nordisk's Ozempic, Wegovy
Fierce Pharma
MAY 31, 2023
FDA warns patients about compounded versions of Novo Nordisk's Ozempic, Wegovy kdunleavy Wed, 05/31/2023 - 13:02
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Fierce Pharma
MAY 31, 2023
FDA warns patients about compounded versions of Novo Nordisk's Ozempic, Wegovy kdunleavy Wed, 05/31/2023 - 13:02

Pharmacy Times
APRIL 7, 2023
Part 1 of this 2-part series examines USP and revisions for sterile and nonsterile compounding, veterinary compounding, FDA inspections of 503A pharmacies, and the interstate distribution of compounded prescriptions post Wellness Pharmacy v.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

STAT
JULY 26, 2023
One of the largest compound pharmacy operations in the U.S. For example, there were 100 instances between November 2022 and March 2023 in which the company found what the FDA called a “significant number” of bacteria, yeast, mold, and other spore-forming organisms. facility run by Central Admixture Pharmacy Services.
Drug Topics
JUNE 15, 2023
Novo Nordisk is the only company with FDA-approved products containing semaglutide.
Pharmacy Times
SEPTEMBER 13, 2022
Prescribers signed a letter expressing concerns about recommendations for restrictions on compounded hormones, which millions of women and other patient populations rely on.

STAT
OCTOBER 12, 2022
Food and Drug Administration and a trade group that represents large compound pharmacies have resolved a long-running dispute over the use of bulk ingredients, clearing the way for some compounded medicines to become available more quickly. Continue to STAT+ to read the full story…

STAT
MAY 15, 2023
At the urging of the Alliance for Pharmacy Compounding, the trade association I lead, the Food and Drug Administration in April 2020 issued temporary guidance allowing traditional compounding pharmacies, within tight regulatory guardrails, to prepare 13 Covid drugs from pure ingredients to meet hospitals’ urgent need.

STAT
NOVEMBER 30, 2023
Novo Nordisk filed lawsuits against two compounding pharmacies for selling adulterated and misbranded compounded drugs claiming to contain semaglutide, the active ingredient in Wegovy and Ozempic (see here and here ). Testing of compounded drugs from these pharmacies revealed unknown impurities up to 33%, the company claims.

Compounding Pharmacy of America
NOVEMBER 13, 2023
Certain patients may need specific treatment that requires compounded medications. Learn more about the top compounded medications. The post Top Ten Medications That Can Be Compounded appeared first on The Compounding Pharmacy of America.
Fierce Pharma
JUNE 20, 2023
Three weeks after the FDA warned that some pharmacies are making unauthorized versio | Novo Nordisk filed five lawsuits in four states on Tuesday, charging health spas, clinics and pharmacies with false advertising, trademark infringement and unlawful sales of compounded versions of diabetes and weight loss drugs Ozempic and Wegovy.

STAT
SEPTEMBER 20, 2023
… As the gold rush over weight-loss drugs accelerates, Eli Lilly filed lawsuits against several compounding pharmacies, spas and wellness centers around the U.S. The drug company accused four compound pharmacies of violating state laws because those versions were not approved by the U.S.

BuzzRx
OCTOBER 14, 2022
If your doctor’s office has given you a prescription with instructions to fill it at a compounding pharmacy, you might be wondering what exactly this means. Only around 7,500 are compounding pharmacies, of which only 3,000 perform sterile compounding. What does it mean when a pharmacy is compounding?

Outsourcing Pharma
OCTOBER 20, 2023
Feedback has been submitted to the US Food and Drug Administration (FDA) with guidelines intended for sponsors of clinical trials related to psychedelic compounds.

Drug Topics
FEBRUARY 10, 2023
The FDA has updated its compounding guidelines for licensed pharmacies, hospitals, and health systems.

Fierce Pharma
JULY 7, 2023
In late May, the FDA warned of illegal knockoffs of Novo Nordisk’s Ozempic and Wegovy. As demand skyrockets for GLP-1 drugs that can trigger significant weight loss, unauthorized versions of the treatments have started to fill pharmacies. | And now, the Danish company has filed its second wave of lawsuits against pharmacies in the U.S.

The FDA Law Blog
FEBRUARY 28, 2023
THC acetate esters are different than delta-9-THC, the psychoactive compound found naturally in the cannabis plant. Dr. Boos explained that delta-9-THCO and delta-8-THCO have similar chemical structures and pharmacological activities as compounds in the cannabis plant so they fall within the definition of THC. Terrence L. Boos, Ph.D.,
STAT
DECEMBER 7, 2023
Not long after the patent was awarded, the company leveraged federally funded research and began investigating different forms of the compound, called Revlimid. But like any pharmaceutical company with an eye toward the future, Celgene continued to seek other ways to wring profits from its development work.

STAT
FEBRUARY 3, 2023
The dietary supplement industry has been begging for federal oversight of products containing cannabidiol (CBD), a non-psychoactive compound found in the cannabis plant that is being incorporated into a bewildering range of oils, tinctures, edibles, and other products. But the FDA turned down industry trade groups , and for good reason.

Pharmaceutical Technology
AUGUST 7, 2023
A new FDA guideline gives manufacturers suggestions to identify carcinogenic drug compounds through clinical study.

Pharmafile
FEBRUARY 22, 2023
Beckley Psytech, a private, clinical-stage biotech company, announced that the FDA has granted Investigational New Drug (IND) approval for their global multi-site phase 2b study into psychedelic drug BPL-003. BPL-003 is indicated for use in patients with Treatment Resistant Depression (TRD). read more

The Checkup by Singlecare
MARCH 25, 2024
Unlike Ozempic, which is FDA approved to treat Type 2 diabetes, Wegovy is approved for weight loss in patients with a body mass index (BMI) of 30 or higher and in patients with a BMI of 27 or higher who also have a weight-related medical condition like Type 2 diabetes, high blood pressure, or high cholesterol.
Pharmaceutical Technology
OCTOBER 31, 2022
The US Food and Drug Administration (FDA) has accepted Outlook Therapeutics’ Biologics License Application (BLA) filing for ONS-5010 / LYTENAVA (bevacizumab-vikg) to treat wet age-related macular degeneration (wet AMD). Outlook stated that ONS-5010 will get 12 years of regulatory exclusivity in the US if it is approved by the FDA.
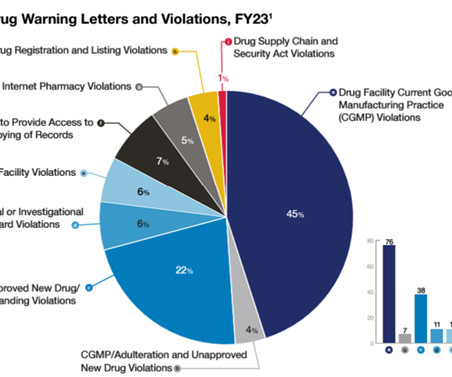
The FDA Law Blog
JANUARY 22, 2024
Some takeaways on those efforts that we gleaned from the report: Compounding. OC devoted a lot of resources to its compliance efforts directed at human drug compounding. This is another area in which FDA works with industry to try to ensure voluntary compliance from regulated entities. Drug Supply Chain. Clinical Trials.

STAT
JULY 20, 2023
The tornado that struck parts of North Carolina on Wednesday not only damaged the immediate area, but could soon make it much harder for countless people across the U.S. and around the world to obtain needed medicines the next time they visit a hospital. Among the damaged buildings was a Pfizer plant in Rocky Mountain, N.C. hospitals.

DiversifyRx
APRIL 4, 2024
Help your patients access the hottest weight loss medications with these semaglutide and tirzepatide resources for independent pharmacy owners, whether you compound or not. Is It Legal To Compound Semaglutide and Tirzepatide? Some states might have you check the list every day you compound with either of these APIs (i.e.,
European Pharmaceutical Review
MARCH 11, 2024
In a review on fungal-contaminated compounded pharmaceuticals and medical devices, researchers have described how the contamination of these products can be due to breaches in sterile compounding procedures. Ahmed et al. One case of drug contamination from 2021 was highlighted in the paper.

The FDA Law Blog
JULY 25, 2023
Recent industry comments submitted to FDA and new, international efforts against these nefarious, potentially carcinogenic organic compounds have the shifting state of regulation here back in the news. What is FDA doing about NDRSIs? Seek, and ye shall find, so FDA hopes. By John W.M. Claud & Sara W.

The FDA Law Blog
AUGUST 8, 2023
Nitrosamines are hardly ever good, Now FDA has issued policy. All of these deviations discussed in FDA inspectional observations and Warning Letters have caused serious issues for manufacturers of APIs or finished drug products. An earlier FDA guidance, revised in 2021. An earlier FDA guidance, revised in 2021.

The FDA Law Blog
JULY 5, 2023
Palmer — Last week FDA published a long-awaited Draft Guidance for outsourcing facilities addressing the Prohibition on Wholesaling Under Section 503B of the Federal Food, Drug, and Cosmetic Act (Draft Guidance). Richardson & Karla L. See 21 U.S.C. 353b(a)(8). Section II at 2.

The FDA Law Blog
AUGUST 14, 2023
OAI is FDA’s classification for facilities it deems to be in an unacceptable state of compliance. One inspection resulted in FDA’s securing a consent decree to restrict or shut down operations, and two resulted in an “untitled letter,” which is not available to the public on FDA’s website.

European Pharmaceutical Review
JANUARY 29, 2024
The guideline states that compounds “at or above the AET must be reported for toxicological risk assessment.” The paper highlighted that SHS analysis is currently a supplementary technique for volatile analysis.

European Pharmaceutical Review
OCTOBER 9, 2022
Research published in the journal Nature Chemistry , shows how researchers from Stanford University in the US uncovered a promising new method to synthetically manufacture the compound tigilanol tiglate, named EBC-46, which could offer targeted medicine for cancer and other diseases.

The Honest Apothecary
APRIL 24, 2023
CALIFORNIA CONSIDERS CLASSIFYING FLAVORING AS “COMPOUNDING “ In a nearly unprecedented move, the state of California is considering treating “flavoring” of a prescription (such as a child’s antibiotic) as “compounding”, thus making the process more difficult and burdensome to pharmacies.

PharmaShots
MARCH 3, 2023
Shots: The US FDA has accepted the NDA for Priority Review of ADX-2191 (methotrexate injection) for PVRL. The therapy received ODD from the US FDA for PVR, PVRL, and retinitis pigmentosa Ref: Aldeyra | Image: Aldeyra Related Post:- Aldeyra Therapeutics Reports the US FDA Acceptance of NDA for Reproxalap to Treat Dry Eye Disease
STAT
NOVEMBER 18, 2022
Diabetic patients will now have easier access to insulin after the FDA approved an Eli Lilly biosimilar as interchangeable with the biologic drug , Bloomberg News notes. Rezvoglar, The agency issued 62 warning letters and 23 import alerts related to drugs, excluding compound-related actions. As of 2019, about 1.9

STAT
JANUARY 25, 2024
One person also experienced hypoglycemia in 2023 after injecting a compounded version of Ozempic, said the organization, which represents 55 regional poison centers across the country and works with the U.S. … Three people sought medical treatment for dangerously low blood sugar in the U.S.

Pharmafile
FEBRUARY 21, 2023
Published date: 21/02/2023 Summary: ● This is the first time the FDA has given IND approval for a Phase IIb study of a short-acting psychedelic like 5-MeO-DMT (Mebufotenin). The study will take place across 40 investigator sites in 7 different countries, with study results expected in 2024. read more

The Checkup by Singlecare
MARCH 7, 2024
Yet the Food and Drug Administration (FDA) hasn’t approved this GLP-1 agonist injection for weight loss. Instead, it’s FDA approved to improve glycemic control in adults with Type 2 diabetes and to reduce the risk of adverse cardiovascular events in adults with Type 2 diabetes and known heart disease.

European Pharmaceutical Review
DECEMBER 13, 2022
They provided this compound to John Hopkins University who received a grant of nearly $4 million from the National Institutes of Health (NIH) for their smoking cessation study in the US – the first time in 50 years the US government has funded a study evaluating a psychedelic compound for therapeutic use.

Omnicell
JANUARY 10, 2023
Sterile compounding is also one of the costliest functions in pharmacy operations. Still, many hospital pharmacies continue to struggle to improve safety and efficiency in their sterile compounding operations. Insourcing IV compounding services at Moses H. Studies have shown that one out of every ten can put patients at risk.

European Pharmaceutical Review
DECEMBER 21, 2022
Although some psychedelics have a long history in traditional medicine, regulatory agencies must evaluate psychedelic compounds the same way they assess any other drug. Psychedelic compounds are assessed by regulators the same way as any other drug. This US pathway offers early-phase benefits, including greater FDA collaboration.

European Pharmaceutical Review
JANUARY 23, 2023
There are major concerns about microbial contamination in cannabis, US Food and Drug Administration (FDA) researchers observed in a study. Investigational New Drug (IND) applications for cannabis as therapeutics tested in clinical trials must comply with FDA’s requirements and standards for drug products. Sopovski et al.
STAT
SEPTEMBER 20, 2022
over allegations that the compound contains a probable carcinogen. … Haleon believes it is not liable for any claims that may arise from U.S. litigation over the Zantac heartburn drug Zantac , Reuters reports. More than 2,000 legal cases related to Zantac have been filed in the U.S.
European Pharmaceutical Review
MAY 7, 2024
percent compound annual growth rate (CAGR) between 2024 to 2029. Approvals in the injectable drug delivery market In Nov 2023, the US Food and Drug Administration (FDA) approved the first and only obesity medication, Eli and Lilly’s Zepbound injection. The market is anticipated to value $1139.4 billion by 2029.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content