Clinical Overview: Lecanemab Use, Coverage for Alzheimer Disease
Pharmacy Times
SEPTEMBER 26, 2023
In clinical trials, lecanemab showed a reduction of amyloid beta plaque in patients with Alzheimer disease.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 clinical alzheimer-disease
clinical alzheimer-disease 
Pharmacy Times
SEPTEMBER 26, 2023
In clinical trials, lecanemab showed a reduction of amyloid beta plaque in patients with Alzheimer disease.
Pharmacy Times
NOVEMBER 17, 2022
Patients with Alzheimer disease administered gantenerumab did experience slower clinical decline, however, it was not deemed statistically significant.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharmacy Times
OCTOBER 11, 2023
Aducanumab, lecanemab, and donanemab are novel monoclonal antibodies designed to slow disease progression and hopefully have a positive impact in the field of Alzheimer disease management.
Pharmacy Times
OCTOBER 17, 2023
Lecanemab is a humanized immunoglobulin gamma 1 monoclonal antibody that targets amyloid plaques, which are thought to play a role in the development and progression of Alzheimer disease.

Pharmacy Times
NOVEMBER 7, 2023
In May 2023, the FDA approved brexpiprazole as the first and only drug to receive approval for treatment of agitation associated with Alzheimer Disease dementia.

Pharmacy Times
JANUARY 8, 2024
Those who were exposed to the clinically acquired stomach bacteria had a 24% increased risk of Alzheimer disease after a decade of onset.
Pharmacy Times
NOVEMBER 3, 2022
Eisai plans to file for traditional approval in the United States by March 31, 2023.

STAT
DECEMBER 18, 2023
When faced with a loved one’s progressive neurodegenerative disease, like Alzheimer’s, or your child’s rare respiratory disease, you question why researchers and resource-backed pharma cannot bring a drug to market quickly enough to help your loved ones.
STAT
JANUARY 31, 2024
Biogen is giving up its ownership of Aduhelm , the Alzheimer’s disease treatment whose 2021 approval led to scrutiny and outrage, turning the page on a tempestuous chapter in the company’s long history.

STAT
OCTOBER 30, 2023
Cassava Sciences has long claimed its experimental drug, called simufilam, slows the cognitive decline of people with Alzheimer’s. On Friday, we learned how: The company recruited a large number of people into its clinical trial who don’t have Alzheimer’s.
STAT
OCTOBER 25, 2023
A version of the Alzheimer’s disease drug Leqembi that can be given with a simpler, under-the-skin injection is equally effective as the currently approved intravenous infusion, according to study results presented Wednesday by its maker, Eisai. Continue to STAT+ to read the full story…
STAT
NOVEMBER 28, 2023
Since the FDA’s approval of lecanemab (marketed as Leqembi) and Medicare’s recent decision to cover the drug, I have met with Alzheimer’s patients and their loved ones who are anxious to know whether they may benefit from this new treatment. Read the rest…
STAT
DECEMBER 2, 2022
SAN FRANCISCO — Scientific meetings about Alzheimer’s disease can be funereal affairs, with researchers from around the world gathering in hopes that the latest in a long line of negative clinical trials might light the path to a long-awaited success. This year was different.

The Checkup by Singlecare
NOVEMBER 14, 2023
Type 3 diabetes is a term used by some researchers to refer to emerging data that shows a link between insulin resistance and dysfunction of a hormone called insulin-like growth factor (IGF-1), which may be a causal factor in Alzheimer’s disease. Here is what we know so far about the connection between blood sugar and dementia.
Hospital Pharmacy Europe
JULY 18, 2023
Donanemab has been found to slow the progression of early symptomatic Alzheimer’s disease after 76 weeks of treatment, according to the findings of a phase 3 randomised trial. Nearly half of those given the drug showed no signs of disease progression after 12 months. women) with a mean age of 73.0 years were recruited.

STAT
JULY 17, 2023
The benefits and risks of Eli Lilly’s treatment for Alzheimer’s disease were confirmed in detailed scientific disclosures Monday, paving the way for its full approval but leaving physicians with lingering questions about the medicine’s impact on patients as a new era in Alzheimer’s therapy unfolds.

STAT
JUNE 9, 2023
Expert advisers to the Food and Drug Administration on Friday voted unanimously in favor of expanding the approval of Leqembi, an Alzheimer’s disease treatment from Eisai and Biogen, further clearing the way for what could be the first widely available medicine that delays the disease’s progress.
STAT
OCTOBER 13, 2023
Medicare has officially expanded its coverage policy for brain scans that detect a brain plaque associated with Alzheimer’s disease, the agency announced on Friday. Previously, Medicare limited coverage for the drug to patients enrolled in clinical studies, and only covered one scan in a patient’s lifetime.
STAT
NOVEMBER 29, 2022
SAN FRANCISCO — A closely watched new treatment for Alzheimer’s disease held up to scrutiny in a detailed scientific presentation Tuesday, as its developers, partners Eisai and Biogen, begin the lengthy process of turning this medicine into what they hope could be a groundbreaking therapy.

STAT
MAY 3, 2023
The conversation on Wednesday centered around decades of evidence linking herpes simplex virus type 1, or HSV-1, to Alzheimer’s. The conversation on Wednesday centered around decades of evidence linking herpes simplex virus type 1, or HSV-1, to Alzheimer’s. Read the rest…

STAT
AUGUST 31, 2023
Alzheimer’s disease has killed at least three women in Astrida Schaeffer’s family over two generations, leaving her feeling helpless and bereft. Last year, the disease killed Schaeffer’s mother’s sister, a former lab technician for Johnson & Johnson. She died in 2019.
European Pharmaceutical Review
DECEMBER 8, 2023
Cerevel’s pipeline includes multiple clinical-stage and preclinical candidates with potential across several diseases including schizophrenia, Parkinson’s disease, and mood disorders. Gonzalez, chairman and chief executive officer of AbbVie. AbbVie also agreed a $10.1

STAT
SEPTEMBER 29, 2022
News of the first clearly successful clinical trial for a new Alzheimer’s disease treatment in two decades has brought hope, scrutiny, and skepticism to a field accustomed to disappointment. Continue to STAT+ to read the full story…

pharmaphorum
MARCH 4, 2022
With prospects of an early, accelerated approval for Roche’s Alzheimer’s disease candidate gantenerumab looking diminished, the drugmaker has launched a new four-year trial in the hope of building its case for the drug. It will now wait for the results of the phase 3 TRAILBALZER-ALZ 2 next year.

STAT
JULY 6, 2023
The Food and Drug Administration on Thursday granted full approval to the first therapy for Alzheimer’s disease clearly shown to slow the cognitive decline associated with the disease — a milestone in treatment, even if the benefits are modest. Continue to STAT+ to read the full story…

STAT
OCTOBER 17, 2022
The next big Alzheimer’s disease study readout is almost here. Roche is nearing the completion of a pair of clinical trials involving its experimental treatment called gantenerumab. The Swiss pharma giant will announce results from the studies within the next six weeks. Continue to STAT+ to read the full story…

STAT
NOVEMBER 13, 2022
Roche reported negative results Monday from a pair of clinical trials investigating an antibody treatment for people with early-stage Alzheimer’s disease — another setback for the Swiss pharma giant’s decades-long effort to develop medicines against the memory-destroying disease.

STAT
MAY 4, 2023
What’s the future of Alzheimer’s disease treatment? Eli Lilly’s investigational medicine for Alzheimer’s met its goals in an all-important clinical trial, and we explain the results, their implications, and the backstory of what could be a blockbuster drug. How do you pivot from a pandemic?

STAT
OCTOBER 17, 2023
The Food and Drug Administration should halt Cassava Sciences’ ongoing clinical trials in Alzheimer’s disease. The FDA has the power to place clinical holds on experimental drugs when there’s evidence of an unacceptable safety risk to study participants. Simufilam doesn’t appear to be toxic.

pharmaphorum
MARCH 15, 2022
A group of pharma companies and academic groups have joined forces with the Digital Medicine Society (DiMe) to develop standardised, digital clinical measures that can be used in the development of therapies for Alzheimer’s disease and related dementias.
STAT
SEPTEMBER 28, 2022
An investigational Alzheimer’s disease treatment from Biogen and Eisai slowed the rate of cognitive decline by 27% in a clinical trial, the companies said Tuesday, meeting the goals of a closely tracked study and strengthening the drug’s case for approval as early as January.
European Pharmaceutical Review
OCTOBER 5, 2023
Longeveron’s Phase IIa trial of its stem-cell therapy for mild Alzheimer’s has revealed in top-line results, that certain doses enabled numerical slowing/prevention of disease worsening relative to placebo. Study safety data were consistent with an established safety profile.

Pharmaceutical Technology
APRIL 6, 2023
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has granted ADvantage Therapeutics’ immunotherapy AD04 an Innovation Passport for the treatment of Alzheimer’s disease. The difficulty in finding a treatment for Alzheimer’s disease has plagued the drug industry for decades.
European Pharmaceutical Review
JULY 17, 2023
Eli Lilly and Company’s TRAILBLAZER-ALZ 2 study has shown donanemab enabled nearly half of participants at earlier stage of Alzheimer’s to experience no clinical progression at one year, according to full results from the Phase III trial. Approximately half of participants met this threshold at 12 months.

pharmaphorum
OCTOBER 12, 2021
A data-mining study conducted by researchers in the US has found that an already-approved diuretic drug could have potential as a treatment for some patients with Alzheimer’s disease. The results are strong enough to back a proof-of-concept study in people with genetic risk of Alzheimer’s according to the researchers.
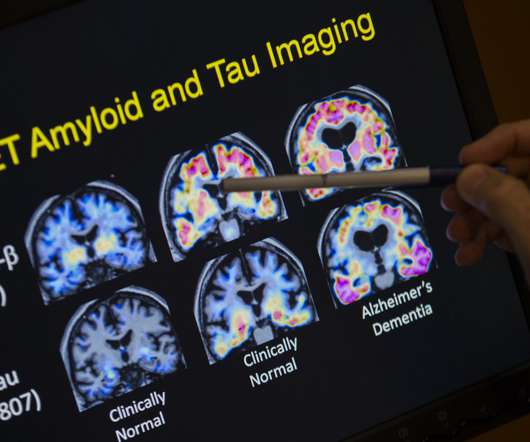
STAT
OCTOBER 6, 2022
Telehealth company Ro has reached an agreement with the National Institute on Aging to create a registry of patients that could participate in Alzheimer’s disease research, a new approach to tackle the stubborn issue of recruitment for clinical trials. Continue to STAT+ to read the full story…
pharmaphorum
DECEMBER 17, 2021
The EMA’s human medicines committee has said it cannot approve Biogen and Eisai’s Alzheimer’s disease therapy Aduhelm, saying it was unconvinced by the data submitted for the drug. The post Biogen’s Alzheimer’s drug Aduhelm is rejected by EU advisors appeared first on.

STAT
DECEMBER 22, 2022
The latest Alzheimer’s disease treatment from Eisai and Biogen needs to be cheaper than $20,000 a year to be cost-effective, according to a draft analysis from an influential nonprofit organization published Thursday. Read the rest…
pharmaphorum
SEPTEMBER 29, 2022
Biogen and Eisai have resurrected hopes that amyloid-targeting drugs could have a benefit in Alzheimer’s disease with a claim that their new drug lecanemab showed a “highly statistically significant” reduction in clinical decline in a phase 3 trial.
pharmaphorum
JUNE 16, 2022
The litany of failed trials of amyloid-targeting drugs for Alzheimer’s disease has added another verse, as Roche concedes that its crenezumab candidate was unable to slow cognitive decline in patients with an inherited form of the disease.

European Pharmaceutical Review
JANUARY 11, 2023
Eisai has submitted a Marketing Authorisation Application (MAA) for lecanemab, for early Alzheimer’s (mild cognitive impairment due to Alzheimer’s (AD) and mild AD dementia) patients in Europe. Clarity AD study for Alzheimer’s. This application was submitted to the European Medicines Agency (EMA).
STAT
SEPTEMBER 29, 2022
The news Tuesday night that a clinical trial of an experimental Alzheimer’s therapy had succeeded hit like a blast — at last, a rare win in a disease devastating nearly 6 million Americans and countless more caregivers. That finding was based on a.45-point Read the rest…
pharmaphorum
OCTOBER 6, 2021
Pfizer’s tyrosine kinase inhibitor Inlyta is already a cornerstone of treatment for some forms of cancer, but could have potential as a therapy for Alzheimer’s disease as well, according to scientists in Canada. The post Could Pfizer’s Inlyta have a role in Alzheimer’s disease?

Pharmaceutical Technology
FEBRUARY 19, 2024
Tonmya is under clinical development by Tonix Pharmaceuticals Holding and currently in Phase I for Alzheimer's Disease.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content