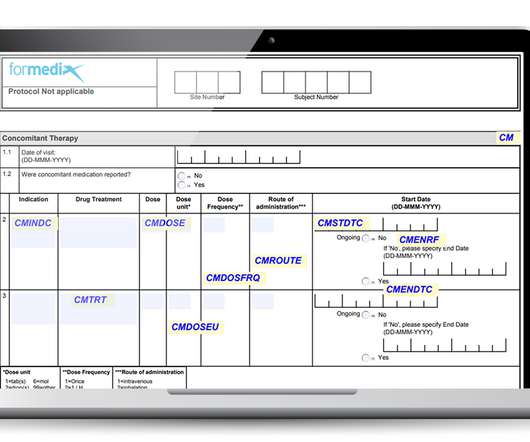
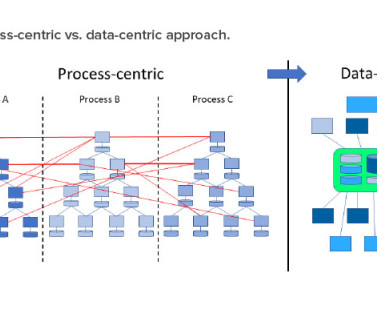
Important factors to consider when working with CRFs
pharmaphorum
AUGUST 10, 2020
Well-designed forms must: Gather data that’s complete, accurate, and of high quality. Be unambiguous and allow for accurate data entry. Avoid gathering more data than what is needed. Provide form completion guidelines to reduce data capture and data entry issues. Avoid duplication. Get user feedback.









Let's personalize your content