Streamlining Postapproval Submissions Using ICH Q12 & SCDM
ISPE
AUGUST 29, 2022
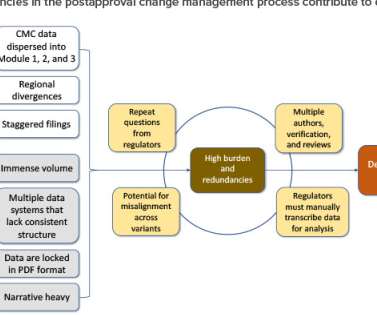
Postapproval change management of pharmaceuticals is an essential part of life-cycle management but is associated with regulatory challenges. In an attempt to address these challenges, in 2019 the ICH endorsed ICH Q12, Technical and Regulatory Considerations for Pharmaceutical Product Lifecycle Management (and Annexes).










Let's personalize your content