FDA pushes back on approving mirikizumab due to manufacturing issues
Pharmaceutical Technology
APRIL 21, 2023
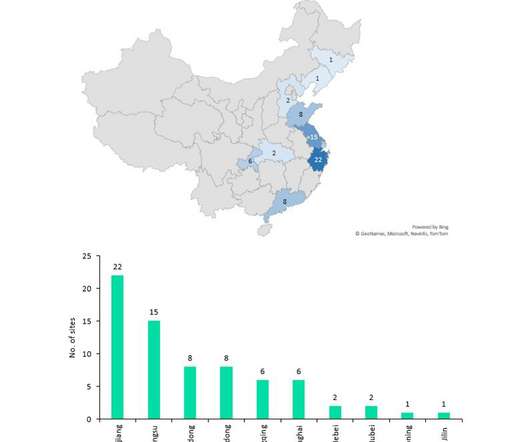
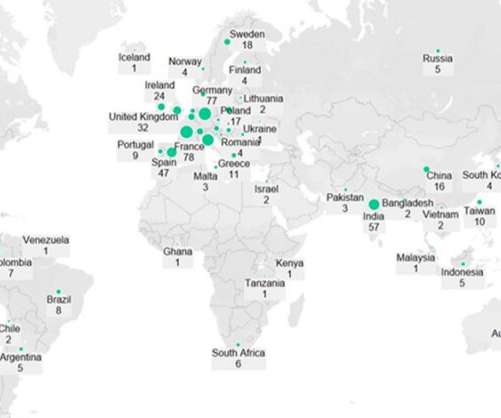
On 14 April, the FDA rejected Lilly’s biologic licence application (BLA) for their anti-interleukin (IL)-23, mirikizumab, which is in development for the treatment of ulcerative colitis (UC). The recent setback dents mirikizumab’s chances to be the first among the IL-23 inhibitors to launch in the US for UC.
















Let's personalize your content