Samsung seeks 'interchangeable' label for Humira biosimilar
pharmaphorum
AUGUST 2, 2023
Samsung seeks 'interchangeable' label for Humira biosimilar Phil.Taylor Wed, 02/08/2023 - 10:37 Bookmark this
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
pharmaphorum
AUGUST 2, 2023
Samsung seeks 'interchangeable' label for Humira biosimilar Phil.Taylor Wed, 02/08/2023 - 10:37 Bookmark this

pharmaphorum
AUGUST 25, 2023
CVS Health launches private label biosimilars unit Phil.Taylor Fri, 25/08/2023 - 08:07 Bookmark this
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

pharmaphorum
NOVEMBER 17, 2023
AZ first to AKT finish line, but FDA clears narrow label Phil.Taylor Fri, 17/11/2023 - 09:46 Bookmark this

pharmaphorum
JUNE 28, 2023
AbbVie, Genmab eye label expansion for Epkinly Phil.Taylor Wed, 28/06/2023 - 13:04 Bookmark this

Drug Store News
JULY 26, 2023
Dollar General is debuting more than 100 new items in its Clover Valley private label line in 2023.

pharmaphorum
JUNE 6, 2023
ASCO: Enhertu data sets up tumour-agnostic label bid Phil.Taylor Tue, 06/06/2023 - 08:26 Bookmark this

Fierce Pharma
MAY 15, 2023
UPDATED: After Supreme Court rejection, Teva mulls options in GSK 'skinny label' feud fkansteiner Mon, 05/15/2023 - 10:59

Hospital Pharmacy Europe
MARCH 26, 2024
It is likely to be safe for penicillin allergy labels (PALs) to be removed for most patients, a study from the University of Birmingham has found. Some 97% of participants labelled with PALs were shown to have no allergy. However, a large body of evidence suggests that 90-95% of PALs are inaccurate.

Drug Store News
JULY 6, 2023
Several beauty trends will be revealed at PLMA’s 2023 annual Private Label Trade Show, Nov. 12-14 in Chicago.

Fierce Pharma
JANUARY 4, 2023
Particulates, label issues prompt separate recalls for Pfizer's Hospira and Accord. Wed, 01/04/2023 - 11:35.

Fierce Pharma
MARCH 20, 2023
Novo Nordisk, Eli Lilly replenish Type 2 diabetes stars Ozempic, Mounjaro after off-label weight-loss boom fkansteiner Mon, 03/20/2023 - 10:09

Fierce Pharma
MARCH 31, 2023
In Teva vs. GSK skinny label feud, Biden admin urges Supreme Court to weigh in fkansteiner Fri, 03/31/2023 - 09:11

Drug Store News
MARCH 23, 2023
The award recognizes the men and women who have significantly contributed to the growth and development of the global private brand industry.

pharmaphorum
MAY 15, 2023
SCOTUS won’t hear ‘skinny label’ lawsuit Phil.Taylor Mon, 05/15/2023 - 14:35 Bookmark this

The FDA Law Blog
DECEMBER 8, 2022
Food and Drug Administration (FDA) issued two guidance documents, one draft and one final, on food allergen labeling requirements. 1, 2023; The applicability of food allergen labeling requirements to specific products (e.g., 1, 2023; The applicability of food allergen labeling requirements to specific products (e.g.,
Pharmaceutical Technology
APRIL 17, 2023
The US Food and Drug Administration (FDA) has approved an update to the indications and usage section of Horizon Therapeutics ’ Tepezza (teprotumumab-trbw) label to specify its use to treat thyroid eye disease (TED) patients regardless of disease activity or duration. The FDA’s approval was granted in January 2020.

The FDA Law Blog
AUGUST 21, 2023
Livornese — I saw the sign…and the answer is no—FDA-approved labeling apparently is not enough under state failure-to-warn laws, according to certain courts. A recent state law failure-to-warn case in the SDNY makes that very point.

OctariusRx
AUGUST 28, 2023
The 2023 version of the OctariusRx medication safety quiz is about to be released. What does the Medication Safety Quiz 2023 cover and how do you get one? The quiz focuses on some of the most commonly encountered situations, including allergies, labeling medications and controlled substances. How are you assessing your staff?

The FDA Law Blog
AUGUST 17, 2023
Tobolowsky — In January 2023, Vanda Pharmaceuticals, Inc. FDA-2023-P-0313 and FDA-2023-P-0344 ) regarding its product Hetlioz (tasimelteon). a)(8)(iv), interpret these provisions to also allow changes due to an aspect of labeling protected by patent or exclusivity. 355(j)(2)(A)(v). 355(j)(2)(A)(v).

Fierce Pharma
FEBRUARY 6, 2024
billion in sales, bringing its 2023 haul to $8.9 On a day when Vertex revealed trial data that indicate | On a day when Vertex revealed trial data that indicate its next-generation cystic fibrosis (CF) treatment will soon be ready to grab the baton, the Boston company also presented figures that show Trikafta remains formidable.

OctariusRx
MAY 15, 2023
Our 2023 nursing calculations quiz is now available. Read the medication label carefully: This a critically important step and one that often leads to errors and potential patient harm. Label all syringes once drawn: Medications that are not for immediate use require proper labeling. Take the quiz and find out!
The Checkup by Singlecare
DECEMBER 6, 2023
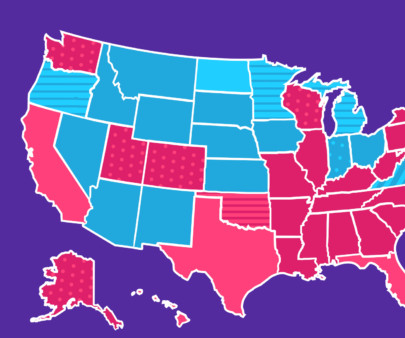
In 2023, the top medications with SingleCare customers included vitamin D supplements, inhalers, and anti-inflammatory drugs. It’s also often prescribed off-label—that is, for a use other than what it’s been approved for—as a treatment for fibromyalgia. Read on to see if any of your prescriptions made the list. Levothyroxine 1.

Fierce Pharma
FEBRUARY 2, 2023
Merck's recent Keytruda label expansion in NSCLC is more of a 'long-term' opportunity, CEO says kdunleavy Thu, 02/02/2023 - 10:43

NF2 BioSolutions
JANUARY 4, 2023
Print our cake labels, bunting and flyers to make your event a great success. The post 2023 Fundraising Challenges appeared first on NF2 BioSolutions. You can set up a personal fundraiser on Peoples fundraising or simply choose the existing NBS UK fundraiser. Buy, bake or ask for donations of cakes.

Fierce Pharma
APRIL 13, 2023
GSK throws counterpunch as 'skinny' label feud with Teva circles Supreme Court fkansteiner Thu, 04/13/2023 - 10:05
The Checkup by Singlecare
DECEMBER 5, 2023
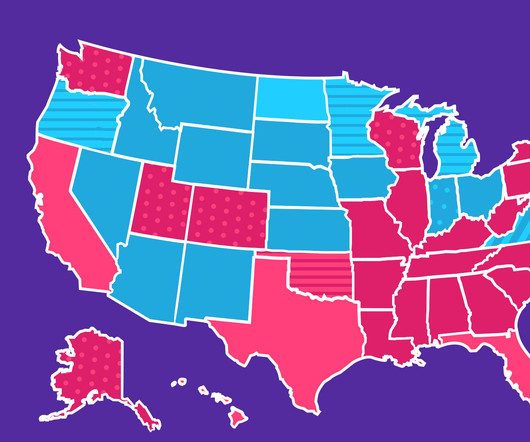
In 2023, nine drugs made our annual list of top prescriptions by state, with vitamin D being the most filled. Keep reading to find out which medications were filled with SingleCare the most in your state in 2023. In 2022 and 2023, it was the most commonly prescribed drug in four states (versus six in 2021 ). People in the U.S.

The FDA Law Blog
MAY 23, 2023
Food and Drug Administration (FDA) released a draft update to its Compliance Policy Guide (CPG) for FDA staff on the Agency’s enforcement of major food allergen labeling and cross-contact. The draft CPG directs FDA field staff to examine possible food product adulteration due to labeling related to allergen cross-contact.

The FDA Law Blog
MARCH 29, 2023
Richardson — Early on March 29, 2023, FDA announced the landmark approval of Narcan (naloxone hydrochloride) Nasal Spray for use as a nonprescription opioid overdose reversal agent. According to this announcement, FDA approval of RiVive is anticipated in July 2023 and the U.S. By Kalie E. launch would be in early 2024.

The FDA Law Blog
FEBRUARY 16, 2023
Richardson — On February 15, 2023, the Nonprescription Drugs Advisory Committee (NDAC) and the Anesthetic and Analgesic Drug Products Advisory Committee (AADPAC) held a joint meeting to discuss an application pending before FDA that would switch Narcan (naloxone) Nasal Spray from prescription to over-the-counter (OTC) status. By Kalie E.

BioPharm
OCTOBER 3, 2023
The new customized system is designed for fully automated and integrated labeling, orientation, and palletization, and it is expected to streamline the current manual processes, according to the press release.

Pharmaceutical Technology
DECEMBER 11, 2023
Novartis is seeking approval for Kisqali as a treatment of early breast cancer, with an FDA application planned by the end of 2023.

PharmaShots
APRIL 20, 2023
treatment interruption/discontinuation due to low lgG & lgM in (0.2% & 10.3%)/ (0.2% & 3.6%) patients Ref: Novartis | Image: Novartis Related News:- Novartis Presents Results of Kesimpta (ofatumumab) in P-III (ASCLEPIOS I/II) and (ALITHIOS) Open-Label Extension Trial for RMS at EAN 2022 CDW, low brain volume change over 5yr.

The Checkup by Singlecare
DECEMBER 6, 2023
In 2023, medications like Ozempic , Mounjaro , and Wegovy soared in popularity—so much so that many pharmacies faced a shortage. Read on to see what readers cared about the most in 2023. Whether you’re taking Ozempic for diabetes or off-label for weight loss, you might be curious if it’s safe to mix it with alcohol.

Drug Topics
APRIL 5, 2023
Off-label drug combinations are increasingly being used in cancer treatments, but there is the potential for overlapping or additive toxicities, explained a speaker at the 2023 HOPA Annual Conference.

European Pharmaceutical Review
OCTOBER 24, 2023
Europe’s pharmaceutical packaging and labelling market is projected to reach $35.78 billion in 2022, the pharmaceutical packaging and labelling market in Europe is expected to expand at a compound annual growth rate (CAGR) of 4.78 percent between the forecast period: 2023 and 2028. percent between the forecast period 2023-2028.
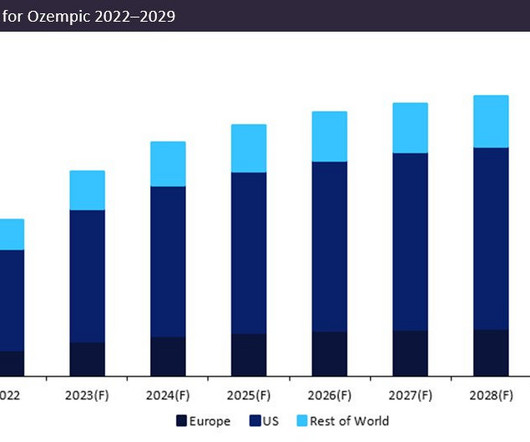
Pharmaceutical Technology
APRIL 28, 2023
Novo Nordisk’s leading drug Ozempic (semaglutide) is forecast to demonstrate a sales growth of 23% in 2023. Ozempic’s forecast 2023 sales of $12.5bn consolidate its position as the dominant market leader, with projected sales in 2023 54% greater than closest competitor Trulicity (dulaglutide) by Eli Lilly, which anticipates sales of $8bn.

PharmaShots
MARCH 17, 2023
5-Adapted Bivalent Booster to Treat COVID-19 in Children ≤5 Years Date: Mar 15, 2023 | Tags: Pfizer, BioNTech, Omicron BA.4/BA.5-Adapted 5-Adapted Bivalent Booster to Treat COVID-19 in Children ≤5 Years Date: Mar 15, 2023 | Tags: Pfizer, BioNTech, Omicron BA.4/BA.5-Adapted

The FDA Law Blog
JANUARY 2, 2023
Labeling in the drug and biologics approval process. FDA Boot Camp is taking place virtually from March 22-23, 2023, visit [link] to learn more! Clinical trials for drugs and biologics. cGMPs and other manufacturing concerns relative to products liability. Proactive adverse events monitoring and signal detection.

ISPE
SEPTEMBER 29, 2023
Locally Focused: Takeda took action to preserve biodiversity on the site and is in the process of obtaining the Réseau Nature label from Natagora, an association for the protection of nature, active in Brussels and Wallonia. Applications must be submitted by 4 December 2023. WANT TO LEARN MORE? Register Now!

PharmaShots
JUNE 16, 2023
Date: June 12, 2023 | Tags: Novartis, Chinook Therapeutics, Atrasentan, Zigakibart, BION-1301, IgA nephropathy, M&A, ~$3.5B Date: June 12, 2023 | Tags: Novartis, Chinook Therapeutics, Atrasentan, Zigakibart, BION-1301, IgA nephropathy, M&A, ~$3.5B

PharmaShots
FEBRUARY 3, 2023
4D Molecular Therapeutics Receives the US FDA’s IND Clearance of 4D-150 for the Treatment of Diabetic Macular Edema Date: Feb 03, 2023 | Tags: 4D Molecular Therapeutics, 4D-150, Diabetic Macular Edema, Regulatory, US, FDA, IND AstraZeneca and Amgen Receive the US FDA’s Approval of Tezspire (tezepelumab) for the Treatment of Severe Asthma (..)

PharmaShots
APRIL 21, 2023
Oblato Reports the First Patient Enrolment of OKN-007 in the P-I Clinical Trial for Recurrent High-Grade Glioma Date: Apr 21, 2023 | Tags: Oblato, OKN-007, Recurrent High-Grade Glioma, Regulatory, Henry Ford Health System CARsgen's CT041 Receives the US FDA’s IND Clearance for the Postoperative Adjuvant Therapy of Pancreatic Cancer Date: (..)

PharmaShots
MARCH 23, 2023
88.9%, respectively The decision was also based on real-world data while the label expansion will provide an effective & convenient prophylactic treatment option for patients with a mod. haemophilia A. haemophilia A. haemophilia A with a sev. haemophilia A with a sev. & moderately sev. vs 13.8mos.)

STAT
JANUARY 25, 2024
One person also experienced hypoglycemia in 2023 after injecting a compounded version of Ozempic, said the organization, which represents 55 regional poison centers across the country and works with the U.S. Ozempic and similar diabetes medicines have been increasingly used off label for weight loss.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content