Biogen, Eisai Alzheimer’s drug lecanemab fast tracked by FDA
pharmaphorum
DECEMBER 24, 2021
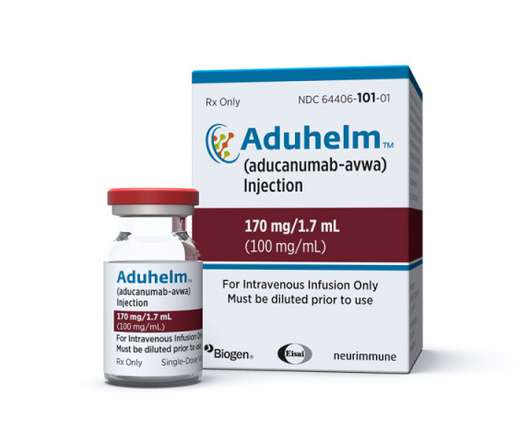
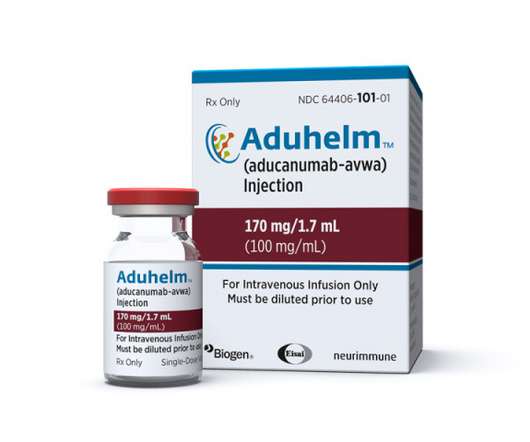
Biogen and Eisai head towards the end of the year with some much-needed good news in their Alzheimer’s programmes, as the FDA awards a fast-track designation to lecanemab, their follow-up to recently approved Aduhelm. The post Biogen, Eisai Alzheimer’s drug lecanemab fast tracked by FDA appeared first on.























Let's personalize your content