Pharmaceutical Executive, December 2022 Issue (PDF)
PharmExec
DECEMBER 8, 2022
Click the title above for a link to open the Pharmaceutical Executive December 2022 issue in an interactive PDF format.

 pharmaceutical-executive-december-2022-issue-pdf
pharmaceutical-executive-december-2022-issue-pdf 
PharmExec
DECEMBER 8, 2022
Click the title above for a link to open the Pharmaceutical Executive December 2022 issue in an interactive PDF format.

ISPE
SEPTEMBER 11, 2023
Herein, DM refers to a manufacturing model in which many units or facilities are disseminated across different geographical areas 6 , 7 DM facilities may produce similar or identical products under a unified pharmaceutical quality system (PQS) or operate as independent units with different outputs. Industry 4.0
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

ISPE
MAY 9, 2023
In an era when the world is accelerating the development of drugs and targeted medicines using innovative technologies, pharmaceutical companies still face registration hurdles for well-characterized molecules because of redundant or additional local regulatory requirements.
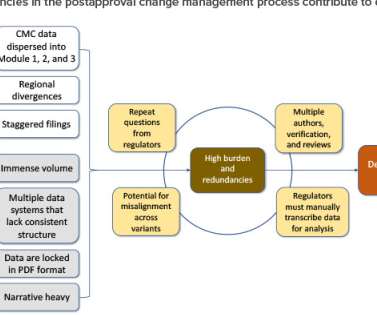
ISPE
AUGUST 29, 2022
Mon, 08/29/2022 - 15:46. September / October 2022. 1 September 2022. Postapproval change management of pharmaceuticals is an essential part of life-cycle management but is associated with regulatory challenges. The European Medicines Agency (EMA) issued its implementation guidance in March 2020. Trudy Patterson.

ISPE
JANUARY 10, 2023
Section 101 of the Defense Production Act of 1950 was extended by Executive Order 13911 in 2020 to Respond to the Spread of COVID-19. Presented at Well Characterized Biotechnology Pharmaceutical (WCBP) Conference. pdf 6 a b c US Food and Drug Administration. Published 13 December 2016. Pharmaceutical/Biotech Man.
Let's personalize your content