3 Critical Keys to Expediting CMC Development
Impact Pharmaceutical Services
JUNE 23, 2023
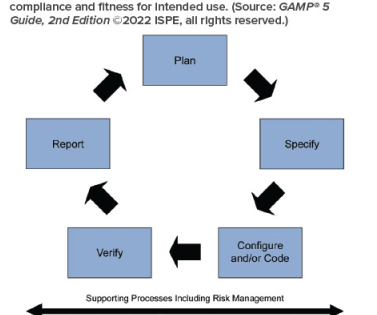
Simultaneously, the creation and use of an SOP manual around the complexities of the QC system ensure uniformity and compliance. SOPs are written instructions for achieving uniformity in specific functions and ensuring compliance among all individuals responsible for these tasks.













Let's personalize your content