A Proposal for a Comprehensive Quality Overall Summary
ISPE
MARCH 29, 2023
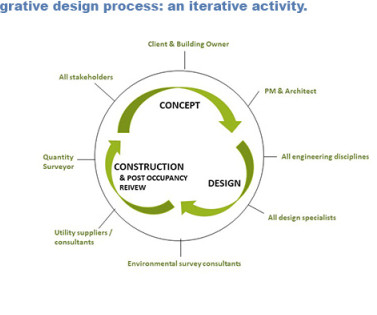
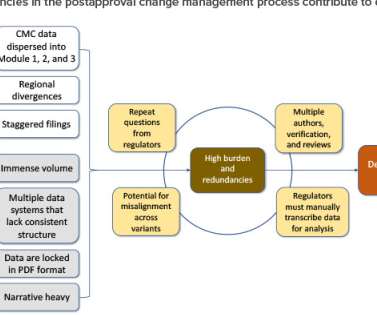
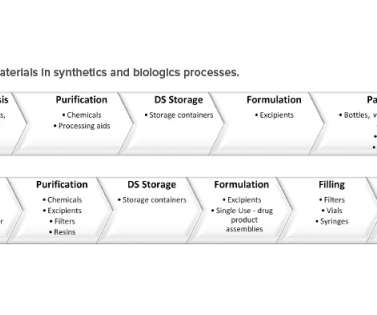
This article summarizes an alternate and more functional way to format the QOS presented in Module 2.3. Currently, it takes time and/or communications (e.g., For purpose of illustration used throughout this article, labels have been assigned to each CQA. Direct feedback from the US FDA on this pilot is shared in the discussion.












Let's personalize your content