FDA Approves GSK's BLA for 5-in-1 Meningococcal Vaccine
Drug Topics
APRIL 16, 2024
The company’s investigational ABCWY vaccine candidate will be reviewed by the FDA by February 14, 2025.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.
Drug Topics
APRIL 16, 2024
The company’s investigational ABCWY vaccine candidate will be reviewed by the FDA by February 14, 2025.
STAT
MARCH 5, 2024
When Americans line up for flu vaccines next fall, they will almost certainly be getting vaccines that no longer contain protection against a family of flu viruses that appears to be extinct.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.
Pharmaceutical Commerce
JANUARY 10, 2024
At the 42nd annual JP Morgan Healthcare Conference, Moderna said new product launches later this year will return the company to sales growth in 2025.

Pharmacy Times
APRIL 16, 2024
The 5-in-1 meningococcal ABCWY vaccine candidate has an assigned Prescription Drug User Fee Act action date of February 14, 2025.

Fierce Pharma
OCTOBER 24, 2023
On the market for two decades, AstraZeneca’s nasal spray flu vaccine, FluMist, may become available for self-administration as soon as next year. | On the market for two decades, AstraZeneca’s nasal spray flu vaccine, FluMist, may become available for self-administration as soon as next year.
Fierce Pharma
AUGUST 23, 2023
In October of last year, GSK laid out a plan to double its business in China by 2025, with much of that expansion dependent upon the succe | In October of last year, GSK laid out a plan to double its business in China by 2025, with much of that expansion dependent upon the success of shingles vaccine Shingrix.

European Pharmaceutical Review
DECEMBER 22, 2022
has finalised a strategic partnership with the UK government to establish a state-of-the-art mRNA vaccine research, development, and manufacturing facility in the UK. It also has potential to develop vaccines for other respiratory diseases, such as flu and respiratory syncytial virus. Moderna, Inc.

Fierce Pharma
JUNE 29, 2023
While Dupixent often steals the show, Sanofi isn’t sleeping on its vaccine franchise. | By 2030, Sanofi figures its immunizations could generate more than €10 billion in annual sales, the company said during a vaccines R&D event Thursday.

European Pharmaceutical Review
OCTOBER 6, 2023
Positive interim results from Moderna’s Phase I/II trial of mRNA-1083 , an investigational vaccine designed to immunise against both influenza and COVID-19, have been released. There are several benefits of the combination vaccine as a preventative measure against these respiratory conditions, according to the pharmaceutical company.

pharmaphorum
DECEMBER 22, 2022
Construction will start early next year of a new manufacturing centre in the UK with the capacity to produce 250 million vaccine doses per year, the centrepiece of a 10-year alliance between the government and US biotech Moderna. The first vaccines are due to be produced at the new facility in 2025.

European Pharmaceutical Review
DECEMBER 19, 2023
Many of today’s vaccines are produced in ready-to-inject liquid formulations that must be kept cold to maintain stability. However, this greatly complicates both the worldwide distribution and stockpiling of vaccines and other drugs. A requisite cold chain has been designed and implemented to be uninterrupted from factory to patient.

STAT
OCTOBER 11, 2023
A company that Pfizer blamed for problems with a clinical trial testing a Lyme vaccine claims that regulators gave its procedures a clean bill of health during a recent inspection. Care Access served as a contract research organization and had enrolled about 3,000 patients in the late-stage trial.
Hospital Pharmacy Europe
OCTOBER 12, 2023
Moderna has announced its combined flu and Covid vaccine has generated strong immunogenicity in early-stage trials. Interim results from its phase 1/2 studies found the vaccine (mRNA-1083) generated antibody levels similar to or greater than flu vaccines as well as similar results to the standalone Moderna Covid jab.

European Pharmaceutical Review
SEPTEMBER 28, 2023
There has been a steady stream of developments in the vaccine manufacturing space throughout 2023. A notable milestone was Moderna’s announcement in April that it had, following finalisation of a ten-year strategic partnership with the UK Government, commenced construction of its mRNA vaccine manufacturing technology centre.

pharmaphorum
DECEMBER 1, 2021
Sanofi has added to its vaccine pipeline with an agreement to acquire Austria’s Origimm Biotechnology and its vaccine-based immunotherapy for people with acne. It will highlight vaccines for pneumococcal disease, meningitis, respiratory syncytial virus (RSV), influenza, and chlamydia. Photo by Scott Webb on Unsplash.

European Pharmaceutical Review
MARCH 6, 2023
” Post-construction, the facility “will harness mRNA science that aims to develop and deliver innovative vaccines to the UK public that address emerging threats from respiratory viruses.” The Moderna Innovation and Technology Centre is expected to become operational in 2025.
pharmaphorum
FEBRUARY 1, 2021
The diplomatic row that broke out last week over the supply of AstraZeneca’s vaccine to Europe appears to have cooled after the UK pharma agreed to supply an extra nine million doses. Step forward on vaccines. The post AstraZeneca to supply nine million more vaccines to EU appeared first on.

European Pharmaceutical Review
APRIL 26, 2023
Built to deliver Moderna’s ten-year strategic partnership with the UK government, announced in March 2023, the MITC will incorporate research, development and a manufacturing facility for mRNA vaccines. Once completed, the MITC is expected to become operational in 2025. Credit: Tom Weller Photography. (L-R)
Pharmaceutical Technology
MARCH 9, 2023
Oxford, UK-based vaccine company SpyBiotech is planning to start dosing subjects for a Phase I trial of its lead vaccine candidate against human cytomegalovirus (HCMV) infection in H2 2023, says chief scientific officer (CSO) Sumi Biswas. The Phase I trial will test three doses of the vaccine, says Biswas.
Pharmaceutical Technology
DECEMBER 6, 2022
Evaxion Biotech and ExpreS 2 ion Biotechnologies have entered a vaccine discovery collaboration agreement to co-develop a new cytomegalovirus (CMV) vaccine candidate. Until 2025, the companies will equally share the research and intellectual property licensing expenses under the partnership.
European Pharmaceutical Review
MAY 14, 2024
This will help support launch of future biologics and vaccines, as well as the continued growth of Dupixent ® ; which could become the first biologic to treat chronic obstructive pulmonary disease (COPD), according to Sanofi. Sanofi shared that it plans to launch in 2025. This investment will support 150 jobs.

STAT
SEPTEMBER 7, 2023
… South Africa was “bullied” into one-sided deals with Covid-19 vaccine suppliers, STAT reports. Vaccine contracts disclosed after a court case between the advocacy group Health Justice Initiative and the South African government showed the country often paid more than other groups for shots. Or something.
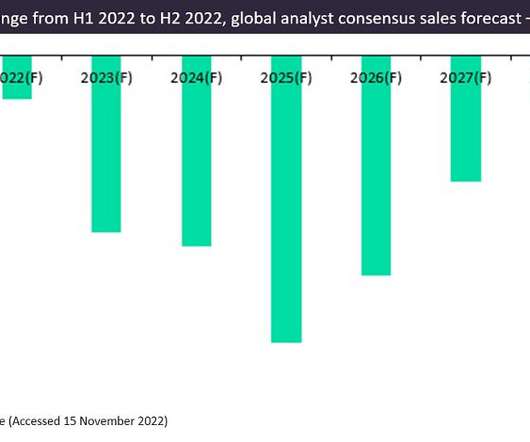
Pharmaceutical Technology
JANUARY 30, 2023
According to GlobalData’s Drugs database, there were 22 prophylactic vaccines in development for COVID-19 with sales forecasts available in H2 2022. Of these 22 vaccines, mRNA-derived vaccines dominate, with Comirnaty and Moderna’s Spikvax accounting for 88% of 2022 sales, with 58% and 30%, respectively.

Pharmaceutical Technology
APRIL 25, 2023
As the prophylactic landscape for malaria has changed in recent years with vaccine approvals, major issues still remain with ensuring access in remote communities. However, making sure the vaccine reaches everyone can be challenging due to the storage requirements. The Mosquirix vaccine contains two vials.

pharmaphorum
SEPTEMBER 15, 2020
billion) order for up to 190 million doses of a coronavirus vaccine in development at French company Valneva. The new deal firms up an earlier agreement in principle with the UK in July that covered up to 100 million doses of VLA2001 , a purified inactivated SARS-CoV-2 vaccine that is expected to require a two-dose vaccination regimen.

Pharmaceutical Technology
DECEMBER 21, 2022
This Moderna Innovation and Technology Centre (MITC) is expected to offer access to a locally produced future mRNA vaccine portfolio against respiratory viruses, subject to regulatory evaluation and licensure. Additionally, the centre is anticipated to have to capacity to make up to 250 million doses of vaccines per year.

European Pharmaceutical Review
JANUARY 31, 2023
A new strategy for 2023-2025 by the Medicines Patent Pool (MPP) aims to reduce access inequities, by improving the availability and affordability of health products in low- and middle-income countries (LMICs). Part of the plan includes enabling 30 million people to access MPP-licensed products each year by 2025.

PQA
JANUARY 12, 2024
Looking ahead, we are excited to announce dates for the 2024 Leadership Summit, as well as the 2025 and 2026 Annual Meetings. Our 2025 Annual Meeting will be in Tampa, Fla., We know you have busy schedules and many meetings to attend throughout the year. This will help you plan far in advance, so that you don't miss any PQA events!

European Pharmaceutical Review
SEPTEMBER 18, 2023
Events, as well as talks from industry leaders and networking opportunities, will be coordinated by a new ‘Scottish Advanced Therapy Medicinal Products (ATMP) and Vaccines Network’ to aid individuals and the wider industry across the north of the UK. Since 2010, the sector has secured eight percent growth each year.

Pharmaceutical Technology
JANUARY 20, 2023
In 2020, Moderna made a net loss of $747 million while its investigational mRNA vaccines were under development. However, during the Covid-19 pandemic, the US-based biotech rose to prominence as it was one of the first companies to develop Covid-19 vaccines with its mRNA technology. billion in Covid-19 vaccine sales.

pharmaphorum
JANUARY 10, 2022
The arrival of COVID-19 brought precious few positives, but one that emerged for the pharmaceutical industry was the emergence and validation of mRNA vaccine technology to prevent disease. Pfizer already has a head start on competitors in the field, having achieved the first US FDA approval for an mRNA vaccine, alongside BioNTech.

PharmaShots
FEBRUARY 19, 2023
The clinical trials remain ongoing as Pfizer continues to enroll new patients at those sites VLA15 is a Lyme disease vaccine candidate that blocks the outer surface protein A (OspA) of the bacteria causing Lyme disease, Borrelia burgdorferi, thereby inhibiting its ability to leave the tick & infect humans.

pharmaphorum
JUNE 29, 2021
Sanofi has just significantly increased its investment in mRNA vaccine development, earmarking €400 million ($475 million) a year in R&D funding to an effort that it hopes will deliver at least six clinical-stage candidates by 2025. . It is also eligible for potential future milestones and other payments up to $1.9

pharmaphorum
DECEMBER 13, 2021
It started with two Covid-19 vaccines – 2021 is the year interest in mRNA therapeutics boomed, reports Katrina Megget. The Covid-19 pandemic showed what could be done with resources and collaboration to accelerate the development of vaccines. There are multiple other mRNA vaccines that are being developed.

Pharmaceutical Technology
JUNE 20, 2023
The proposed institute will focus on vaccine research, public health research, therapeutics, genomics, diagnostics, clinical trials and data. Demolition of the existing buildings on the site will commence next year, and construction work on the new building will begin in 2025.

pharmaphorum
MARCH 8, 2022
Moderna has unveiled plans to start clinical trials of mRNA-based vaccines for 15 infectious diseases that pose the biggest public health risks around the world – including HIV, malaria and tuberculosis – by 2025.

Pharmaceutical Technology
MAY 22, 2023
This collaboration will back the early development of solutions to treat life-threatening drug-resistant infections including new antibiotics, vaccines and rapid diagnostics to prevent death and disease worldwide. The UK has already given £20m from its official development assistance (ODA) budget to the effort.
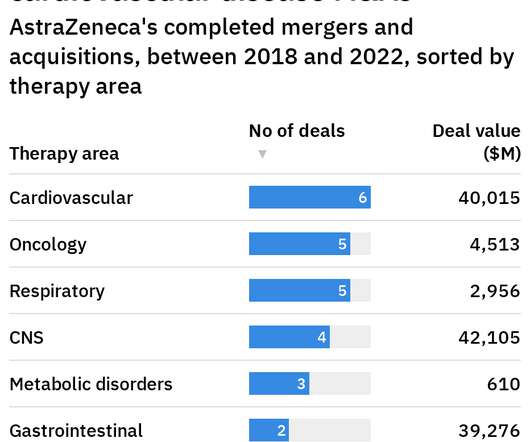
Pharmaceutical Technology
SEPTEMBER 5, 2022
Following AstraZeneca’s success in vaccine development during the Covid-19 pandemic , the pharmaceutical giant is now looking to expand its scope through acquisitions across a range of indications, says CEO Pascal Soriot. “We We want to be a sustainably growing company.

pharmaphorum
JUNE 22, 2022
Moderna has said it will open a new R&D and manufacturing facility in the UK which could lead to improved access by NHS patients to mRNA-based vaccines and drug treatments. Construction of the facility is expected to start later this year, said the government, with the first mRNA vaccines due to be produced there in 2025.
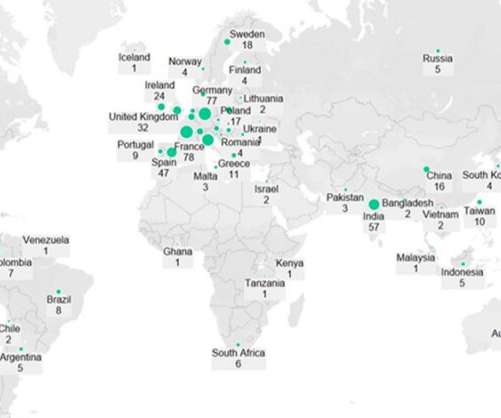
Pharmaceutical Technology
JULY 29, 2022
Pfizer (New York, New York) and Moderna’s (Cambridge, Massachusetts) Omicron-specific Covid-19 vaccines are in late-stage development, have demonstrated good efficacy against the variant and will likely be available to the public later this year to provide an additional booster and increase demand for injectable manufacturing.

Pharmaceutical Technology
MAY 11, 2023
The business continuity plan was introduced in 2020 to allow European regulators to prioritise the assessment of Covid-19 therapeutics and vaccines while also handling other responsibilities. This meant Covid-19 medicines were prioritised when it came to different pharmacovigilance processes.

European Pharmaceutical Review
JANUARY 13, 2023
Priorities for 2022-2025. These included 40 experts appointed by the EPC to join the newly created Working Party on mRNA vaccines , following a call for experts issued in the summer of 2022. European Pharmacopoeia Commission’s November 2022 session highlights: Approval of the Ph. Appointment of 886 experts from Ph. and non-Ph.

The FDA Law Blog
DECEMBER 3, 2023
On November 17th, CMS issued its final guidance on the Discount Program in which it responded to public comments and provided updated guidance for the Discount Program for 2025 and 2026. Manufacturers must sign the agreement by March 1, 2024, to participate in the 2025 plan year. covered insulin product or vaccine).

pharmaphorum
JANUARY 6, 2022
IQVIA Institute models global cumulative spend on COVID-19 vaccines to be a base case of $251bn to 2026 , with spend on COVID-19 treatments, including the new oral agents and treatments of post-viral syndromes, likely to add significant incremental spend. Medicine budget constraint, structural fund largesse.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content