FDA Roundup: Drug Approvals From April 2024
Drug Topics
APRIL 29, 2024
Check out these important FDA updates from the month of April 2024.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

Drug Topics
APRIL 29, 2024
Check out these important FDA updates from the month of April 2024.

STAT
DECEMBER 27, 2023
WASHINGTON — The Food and Drug Administration has a lot on its plate in 2024 with the reorganizations of its food center and the office that inspects FDA-regulated facilities. Amid that massive reorganization, the FDA also will lose one of its most influential leaders.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Drug Topics
AUGUST 25, 2023
The new deadline to be in compliance with the requirements will be November 27, 2024.
European Pharmaceutical Review
APRIL 30, 2024
Last week, the US FDA approved a new gene therapy for eligible adults with haemophilia B. CHMP meeting highlights: March 2024 The post CHMP meeting highlights: April 2024 appeared first on European Pharmaceutical Review.

Pharmafile
NOVEMBER 8, 2023
Johnson & Johnson MedTech have announced plans to submit the Ottava robotic surgical system to the US Food and Drug Administration (FDA) for an investigational device exemption (IDE) application in the second half of 2024. This will allow the company to move its device into US-based clinical trials in 2024.

Pharmacy Times
AUGUST 28, 2023
The start date of the package-level electronic tracking system will shift from November 27, 2023 to November 27, 2024.

STAT
JANUARY 18, 2024
And what’s going to move markets in 2024? We also look ahead to the biggest biotech stories coming in 2024, including some major data readouts and a few pivotal FDA decisions. Why are rich biotechs cutting costs? Has JPM Week already worn off? Read the rest…

European Pharmaceutical Review
MARCH 5, 2024
International participation and trending topics With over 900 exhibitors, analytica 2024 promises an impressive lineup. analytica 2024 provides a comprehensive overview of innovative analytics and molecular – as well as microbiological – testing methods. Top topics at the trade fair include digitalisation, Laboratory 4.0,

PharmaVoice
JANUARY 12, 2024
Following a year where the FDA approved 55 new drugs, the 2024 PDUFA calendar is set with a number of potentially buzzy approvals.

Pharmacy Times
OCTOBER 17, 2023
The committee will help the FDA address issues related to digital health technologies, including artificial intelligence, virtual reality, cyber security, and patient-generated health data.

pharmaphorum
JANUARY 8, 2024
Dr Peter Marks, director of the FDA Center for Biologics Evaluation and Research told a packed house in San Francisco for JPM that FDA wants to support gene therapies in 2024.

OctariusRx
APRIL 8, 2024
Drug shortages currently remain at over 100 different medications, with the Food and Drug Administration (FDA) noting 116 products as being unavailable. The post Drug Shortages Update: April 2024 appeared first on OctariusRX. This week we are once again providing an update on the most recent drug shortages. Contact us for assistance.
STAT
MARCH 5, 2024
Experts who advise the Food and Drug Administration on vaccine-related issues voted unanimously on Tuesday to recommend that the FDA approve trivalent flu vaccines for the 2024-2025 season, instead of the quadrivalent, or four-in-one, shots that have been the industry standard for the past decade or so. Read the rest…

pharmaphorum
MAY 1, 2024
Final FDA rule on LDTs creates a paradigm shift for laboratory manufacturers and partners, announces Avalere Mike.Hammerton Thu, 02/05/2024 - 06:35 Bookmark this

Fierce Pharma
FEBRUARY 14, 2024
FDA unleashes multiple warning letters targeting insanitary manufacturing and online sales of unapproved Mounjaro, Ozempic fkansteiner Wed, 02/14/2024 - 05:10

OctariusRx
JANUARY 22, 2024
Drug shortages currently remain at over 100 different medications, with the Food and Drug Administration (FDA) noting 124 products as being unavailable. The post Drug Shortages Update: January 2024 appeared first on OctariusRX. This week we are once again providing an update on the most recent drug shortages.

Fierce Pharma
MARCH 11, 2024
Viatris likely just lost one of its top product debuts for 2024, thanks to an FDA rejection of its partner Mapi Pharma’s application for a new version of Teva’s multiple sclerosis (MS) drug Co | Viatris likely just lost one of its top product debuts for 2024, thanks to an FDA rejection of its partner Mapi Pharma’s application for a new version of Teva’s (..)

The FDA Law Blog
AUGUST 24, 2023
(“HP&M”) is is proud to announce that 14 of the Firm’s attorneys have been selected to the 2024 edition of The Best Lawyers in America®. Torrente (FDA Law), Michelle L. Butler (FDA Law), Sara W. Koblitz (FDA Law), Allyson B. Mullen (FDA Law), and Anne K. Walsh (FDA Law), as well as McKenzie E.

The Checkup by Singlecare
JANUARY 19, 2024
Food and Drug Administration (FDA) has granted the state of Florida the authority to import certain prescription drugs from Canada—an important measure to counteract soaring prescription costs. After delays that took a lawsuit to resolve, the FDA authorized the program on Jan. In a landmark decision, the U.S. an outlier? an outlier?

pharmaphorum
JANUARY 31, 2024
FDA approval numbers soar after COVID troubles Mike.Hammerton Wed, 31/01/2024 - 12:00 Bookmark this
Pharmacy Times
FEBRUARY 7, 2024
The FDA assigned a Prescription Drug User Fee Act (PDUFA) goal date of October 8, 2024.

European Pharmaceutical Review
MARCH 13, 2024
FDA inspections Identification of data integrity deviations Of the 70 Warning Letters issued by the US Food and Drug Administration (FDA) so far in 2024, three have identified data integrity issues at pharmaceutical manufacturing sites outside the US.
Drug Topics
NOVEMBER 29, 2023
The treatment from Arcutis Biotherapeutics has a Prescription Drug User Fee Act (PDUFA) target action date of July 7, 2024.

Pharmacy Times
APRIL 4, 2024
MDMA-assisted therapy and psilocybin currently lead the way with potential approvals by the FDA.
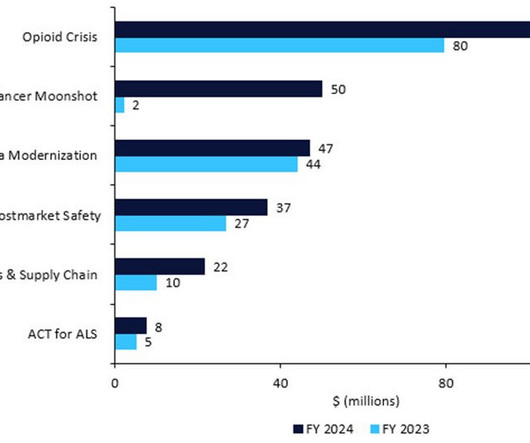
Pharmaceutical Technology
MARCH 30, 2023
The US FDA has revealed its detailed budget proposal for FY2024, which would require pharma companies to name their active pharmaceutical ingredient (API) suppliers, restart President Biden’s Cancer Moonshot, inject cash into amyotrophic lateral sclerosis (ALS) research, and enforce stricter rules around manufacturing, recalls, and shortages.

Pharmaceutical Technology
MARCH 11, 2024
The FDA is still likely to approve donanemab, however, the delay will benefit Eisai and Biogen in allowing Leqembi to gain greater traction in the Alzheimer's market.
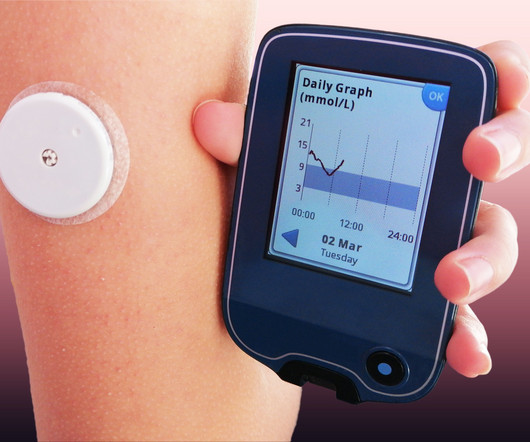
Drug Topics
MARCH 5, 2024
Dexcom's Stelo will be available in summer 2024.
Pharmaceutical Technology
SEPTEMBER 12, 2023
The US FDA has approved Pfizer and BioNTech’s sBLA for Omicron XBB.1.5-adapted adapted monovalent Covid-19 vaccine, COMIRNATY 2023-2024 Formulation.

The FDA Law Blog
FEBRUARY 26, 2024
(HP&M) proudly announces that Director Anne Walsh will be a featured panelist at the highly anticipated 2024 Women’s White Collar Defense Association (WWCDA®) Leadership Retreat and Annual Attorney Meeting. The event is set to take place from March 4-6, 2024, in San Francisco, California. More notably, Ms.

The FDA Law Blog
MARCH 19, 2024
AMDM is known for serving as a “connector” for FDA and other regulatory bodies to share information and exchange ideas with industry. Mullen brings to the AMDM board a wealth of knowledge and expertise gained from her years of providing counsel to medical device and IVD manufacturers. Speaking about her appointment, Ms.

The FDA Law Blog
NOVEMBER 15, 2023
Firms included in the 2024 Best Law Firms ranking are recognized for professional excellence with persistently impressive ratings from clients and peers. To be considered for this milestone achievement, at least one lawyer in the law firm must be recognized in the 2024 edition of Best Lawyers.

Pharmaceutical Commerce
AUGUST 30, 2023
Event’s first session aims to analyze the legislation’s implementation, as FDA enforcement is delayed until November 2024.

Pharmaceutical Technology
DECEMBER 20, 2023
The FDA accepted the filing based on NIH-funded Phase III trial interim results, with an approval decision expected in Q1 2024
STAT
OCTOBER 5, 2023
The Food and Drug Administration’s expert vaccine advisory panel on Thursday unanimously endorsed the idea of taking a strain of influenza viruses that no longer appears to circulate out of flu shots as quickly as possible, pressing the FDA and manufacturers to try to get the work done on an expedited timeline.
Pharmacy Times
MARCH 5, 2024
The continuous glucose monitoring system will be available for purchase online and without a prescription starting in summer 2024.

The Checkup by Singlecare
MARCH 8, 2024
On March 5, 2024, the U.S. Food and Drug Administration (FDA) approved the first over-the-counter (OTC) continuous glucose monitor (CGM). According to DexCom, the Stelo glucose biosensor will be available for purchase online in the summer of 2024. The Dexcom Stelo Glucose Biosensor System is for adults who don’t use insulin.

The FDA Law Blog
DECEMBER 21, 2023
Both are celebrating their 75th anniversaries in 2024. 2024 brings a host of Hyman, Phelps & McNamara, P.C. 2024 brings a host of Hyman, Phelps & McNamara, P.C. (“HPM”) In other exciting news — in 2024 FDLI will be getting a make-over! What do Gene Autry’s Rudolph the Red-Nosed Reindeer and FDLI have in common?

European Pharmaceutical Review
NOVEMBER 2, 2023
The US Food and Drug Administration ( FDA) has approved Wezlana (ustekinumab-auub) as a biosimilar to Johnson & Johnson’s Stelara (ustekinumab). However, he did note that Stelara does have a composition of matter patent expiry in mid-2024 in Europe. US sales of Stelara totalled $6.4
Pharmafile
NOVEMBER 30, 2023
Karuna Therapeutics has announced that the US Food and Drug Administration (FDA) has accepted its New Drug Application (NDA) for KarXT (xanomeline-trospium) for the treatment of schizophrenia in adult patients. The NDA has been given a Prescription Drug User Fee Act (PDUFA) date of 26 September 2024.

The Checkup by Singlecare
MARCH 13, 2024
5, 2024, the U.S. Food & Drug Administration (FDA) approved Jubbonti and Wyost, the first interchangeable biosimilars to the injectable medications Prolia and Xgeva , respectively. For traditional medications, the FDA can approve generic medications. Osteoporosis and bone density conditions are common in the U.S.
Pharmafile
JANUARY 17, 2024
Aruna Bio has announced that the US Food and Drug Administration (FDA) has cleared its Investigational New Drug (IND) application for its lead programme, AB126, meaning it can enter a phase 1b/2a clinical trial for acute ischemic stroke. The trial is expected to begin in the first half of 2024.

Fierce Pharma
SEPTEMBER 12, 2023
Xspray Pharma, before receiving an FDA slapdown in July, had expected to launch its competitor to Bristol Myers Squibb’s leukemia blockbuster Sprycel within the second half of 2023. 1, 2024, or potentially earlier under certain circumstances. 1, 2024, or potentially earlier under certain circumstances. 28, 2026.

Pharmaceutical Technology
MARCH 8, 2024
While Lilly’s Alzheimer’s drug was previously expected in Q1 2024, an FDA-mandated review at an unknown date means a decision may be delayed.

PharmExec
FEBRUARY 21, 2024
The FDA assigned the biologics license application for linvoseltamab to treat relapsed/refractory multiple myeloma with a Prescription Drug User Fee Act of August 22, 2024.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content