Streamlining Postapproval Submissions Using ICH Q12 & SCDM
ISPE
AUGUST 29, 2022
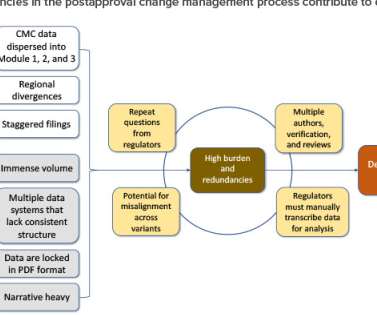
This article will examine the current global regulatory submission workflow for postapproval CMC changes and will propose principles to streamline regulatory submission authoring through the utilization of ICH Q12 concepts, in combination with SCDM. Journal of Pharmaceutical Sciences (2021). Sugrue-Richards, and G. doi:10.1016/j.xphs.2021.09.046.








Let's personalize your content