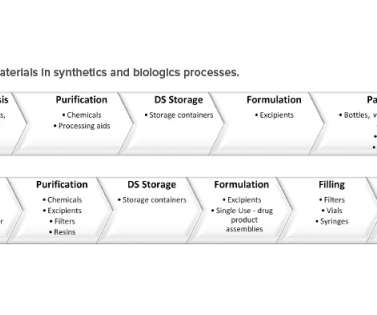
Considerations for a Decentralized Manufacturing Paradigm
ISPE
SEPTEMBER 11, 2023
September 2004. Nature Communications 12, no. Sanofi Pumps $554M into New Vaccine Manufacturing Facility in Eastern France.” www.fiercepharma.com/manufacturing/sanofi-pumps-554m-into-new-vaccine-manufacturing-facility-eastern-france 41 Boccard. New Project: Evolutive Vaccines Facility for Sanofi.” 3 (2020):e2970.









Let's personalize your content