Regulatory Landscape for Raw Materials: CMC Considerations
ISPE
AUGUST 30, 2022
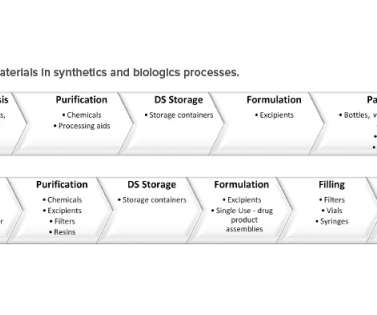
With shortages, regulatory complexity is compounded due to differences in submission and data requirements from various regulatory agencies. In this article, the term “raw material” refers to a material used in the manufacturing and packaging of a drug substance (DS) or a drug product (DP). Published 2002. Burke, PhD.







Let's personalize your content