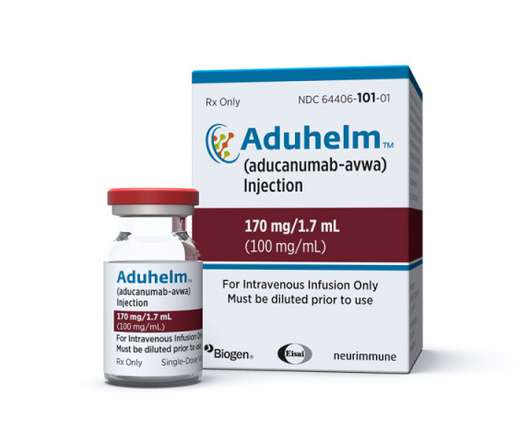
FDA slammed over Aduhelm OK as Biogen takes heat for $56k price tag
pharmaphorum
JUNE 8, 2021
The FDA “has failed in its responsibility to protect patients and families from unproven treatments with known harms” in approving Biogen’s Alzheimer’s disease drug Aduhelm. ICER points to brain swelling and potential bleeding seen in approximately 30% of patients treated with aducanumab.












Let's personalize your content