Cereno partners with CordenPharma to scale up CS1 drug production
Pharmaceutical Technology
JANUARY 12, 2024
Cereno Scientific has announced a partnership with CordenPharma to scale up manufacture of its drug candidate CS1.
This site uses cookies to improve your experience. By viewing our content, you are accepting the use of cookies. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country we will assume you are from the United States. View our privacy policy and terms of use.

 production scale-up
production scale-up 
Pharmaceutical Technology
JANUARY 12, 2024
Cereno Scientific has announced a partnership with CordenPharma to scale up manufacture of its drug candidate CS1.

Fierce Pharma
NOVEMBER 22, 2023
Unable to scale up its manufacturing fast enough to meet the spiraling demand for its GLP-1 weight loss products, Novo Nordisk is employing a new strategy—reducing production of diabetes drug Victo | Unable to scale up its manufacturing fast enough to meet the spiraling demand for its GLP-1 weight loss products, Novo Nordisk is employing a new strategy—reducing (..)
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

European Pharmaceutical Review
MAY 1, 2024
These technologies could enable patients to access advanced treatments up to one year faster, CPI stated. The aim is to support manufacturers to be adaptable during production scale up and navigate the balance between medicine demand and supply.

Pharmaceutical Technology
MARCH 13, 2024
In T-cell manufacturing, the historical issue of maintaining the quality of materials when scaling up production is changing.

Fierce Healthcare
OCTOBER 10, 2023
. | Alongside internal innovation efforts, the California system said the center will be a door for early-stage partners to co-develop and scale their digital health products in real-world clinical environments. Sutter Health said it aims to have the center up and running in San Francisco by early 2024.

BioPharm
NOVEMBER 15, 2023
As oligo manufacturers scale up production from lab-scale to clinical and commercial scales, they may require volumes of hazardous materials that drive additional facility design considerations.

European Pharmaceutical Review
APRIL 11, 2024
This is because they can selectively manipulate processes in the body such as “gene expression, splicing, and protein production”, the companies explained. “Moreover, our involvement from the very early stages of the project allows us to support and facilitate future scale-up efficiently.”

European Pharmaceutical Review
FEBRUARY 3, 2023
Switzerland-based contract development and manufacturing organisation (CDMO) CARBOGEN AMCIS has opened a new facility in France, dedicated to the custom development and production of sterile injectable drug products. The lines offer a flexible and versatile set up, which is unique within Europe.

European Pharmaceutical Review
SEPTEMBER 21, 2023
To establish it, the Novo Nordisk Foundation is committing up to £127 million. A few hospitals in Denmark already have small-scale facilities for manufacturing products for these innovative treatments. A few hospitals in Denmark already have small-scale facilities for manufacturing products for these innovative treatments.

Pharmaceutical Technology
AUGUST 2, 2022
Samsung Biologics and GreenLight Biosciences have completed the initial commercial-scale engineering run for their messenger ribonucleic acid (mRNA) Covid-19 vaccine under their manufacturing collaboration. Furthermore, the mRNA synthesis reaction of GreenLight had a titer of 12g/L at a commercial scale and yielded 650g mRNA.

European Pharmaceutical Review
NOVEMBER 9, 2023
When it comes to biologics, scaling up production to ensure global supply is a remarkable process that embodies Industry 4.0 and the very latest in advanced manufacturing technologies. We also explore opportunities for outsourcing of regulatory activities, the challenges of paediatric drug formulation and much more.

The FDA Law Blog
OCTOBER 27, 2022
This final guidance provides recommendations to original applicants and holders of approved applications for human drugs and certain biological products on implementing chemistry, manufacturing, and controls (CMC) postapproval changes(s) through the use of a Comparability Protocol (CP).

European Pharmaceutical Review
NOVEMBER 24, 2023
billion (16 billion Danish kroner) to expand its production site in Chartres, France, supporting manufacture of its products for serious chronic diseases. Aseptic production and finished production processes will be added to the site, as well as an extension of the current Quality Control Laboratory.

European Pharmaceutical Review
DECEMBER 9, 2022
Catalent, a contract manufacturing organisation (CMO), has opened one of the largest commercial-scale cell therapy manufacturing facilities in the world, at its European Center of Excellence for Cell Therapies in Gosselies, Belgium. Manufacturing facilities at the Gosselies campus . Catalent cell therapy manufacturing.
European Pharmaceutical Review
JUNE 27, 2023
The companies will partner in the process development and manufacturing scale-up at a new, large-scale facility in Portsmouth, New Hampshire, US. Operated by Lonza, the 130,000 square foot facility is anticipated to create up to 300 new jobs at peak capacity.

European Pharmaceutical Review
AUGUST 25, 2022
DURING THE development of new types of active pharmaceutical ingredients (APIs) or medicinal products, scale-up at a pilot plant is an important proof of concept. Water may be found in raw materials, where it can impact blending, granulation and synthetic reactions, as well as intermediate and final products.
European Pharmaceutical Review
MARCH 7, 2024
Last month, construction began on Europe’s first industrial-scale facility dedicated to the production of lead-212 based radioligand therapies. A report from Technavio suggests that due to trends in the nuclear medicine market such as isotope production, the radiopharmaceuticals market will grow by $4.61

ISPE
SEPTEMBER 13, 2023
Project: NISHWAS Six Months to Commercially Scale COVID-19 Vaccines SERUM’s NISHWAS project is recognized as a FOYA Category Winner in both Supply Chain and Social Impact. SERUM’s aseptic and sterile drug substance and drug product facility project was being set up for other vaccines products.

European Pharmaceutical Review
OCTOBER 24, 2022
Three trade bodies representing vaccine creators and manufacturers are endorsing The Berlin Declaration framework , a proposal that outlines how the industry would reserve an allocation of real-time vaccine production for swift distribution to priority populations in lower-income countries in future pandemics.
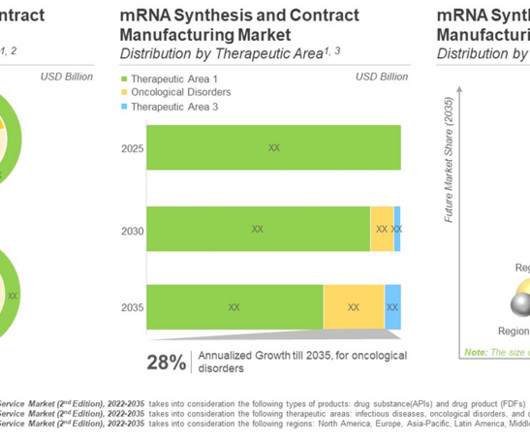
Roots Analysis
AUGUST 16, 2023
The success of COVID-19 vaccines paved the path for mRNA-based drug products. Safety, efficacy and rapid production of mRNA drives the interest in this domain. Concluding Remarks Currently, dozens of preclinical and clinical reports demonstrating the efficacy of these platforms have been published in the last two years alone.

European Pharmaceutical Review
NOVEMBER 16, 2022
For young biotechs, scaling up production brings green-field challenges, while for more established pharma a shift may be required to ensure that facilities are optimised for new ways of working. That is because of the relative expense of emerging therapies and the pressure on pricing for continuing product lines.
Pharmaceutical Technology
SEPTEMBER 7, 2022
Bharat Biotech carried out the product development linked to preclinical safety assessment, large-scale production scale-up, as well as the development of formulation and delivery device, including trials in humans. iNCOVACC has been designed for efficient distribution and easy administration.”.

ISPE
NOVEMBER 2, 2022
The industry is in the process of a transformation that was sped up by the pandemic, which enhanced innovation and collaboration. Cheslock noted the many initiatives that pharmaceutical companies including Pfizer have—top down and bottom up site initiatives—and noted the challenge is to integrate all of these efforts to deliver full impact.

Pharma Mirror
MAY 31, 2023
In the face of increasing global health challenges, the development and production of vaccines have become paramount. One critical aspect is the ability to scale up manufacturing processes to meet the growing demand for viral vector-based vaccines.
European Pharmaceutical Review
AUGUST 23, 2022
Researchers from the University of Arkansas (U of A) and two partner institutions have been awarded a $6 million grant by the US’s National Science Foundation (NSF) to develop purification membranes for future large-scale manufacturing of viral vectors and virus-like particles.

pharmaphorum
APRIL 6, 2022
The growth of a start-up after obtaining Series A funding is a transformational phase when scaling up the company becomes critical. The CEO can experience pressure to deliver on the revenue growth goals, whilst still developing their product, growing the team, and implementing core processes for the company.

BioPharm
MAY 4, 2023
This guide explains how biopharmaceutical production and bioprocessing trends demand flexibility and consistency in R&D, scale-up, and production processes, requiring optimization through analytics.

European Pharmaceutical Review
JULY 7, 2023
Turpentine is also a waste product from the paper industry. Pharma within planetary boundaries Benefits of sustainable turpentine-based pharmaceuticals The researchers hope that this more sustainable , ‘biorefinery’ approach could replace the need for crude oil products in the chemical industry.

European Pharmaceutical Review
MARCH 27, 2024
Overall, this indicates that the Swedish life sciences industry is strengthening its position as a significant player in the global market, with innovation and high-quality product exports specifically helping to drive global demand, SwedenBIO claimed. Most innovative companies may not have the approach to manufacture and sell the product.

European Pharmaceutical Review
FEBRUARY 14, 2024
The development of drug products containing live anaerobic bacterial strains as the active pharmaceutical ingredient and where potency and shelf-life is typically associated with viable cell quantification, poses unique challenges when compared to both small and large molecule development therapeutics.
European Pharmaceutical Review
MAY 16, 2023
Using AI to manufacture biosimilars As part of the strategic partnership, both parties share a commitment “to use disruptive technology with lower operational costs to deliver high-quality biosimilars at scale to patients around the world,” according to Sandoz’s CEO Richard Saynor. “By

European Pharmaceutical Review
MAY 2, 2024
There are a lot of conjugated products in the clinical pipeline and novel conjugation partners and chemistries are being tried. Automated microbioreactors have been in place for many years, and these platforms are being used as validated scale-down models for large-scale bioreactors. How can these be overcome?

European Pharmaceutical Review
AUGUST 29, 2022
Oftentimes, our clients’ goals are to increase cell growth, improve protein titers and optimise product quality attributes. Having scale-up-ready media and processes is a big concern as well. How can custom media be produced at larger scale for use at the manufacturer’s site?

European Pharmaceutical Review
JUNE 27, 2022
According to the ClinicalTrials.gov trial registry, as of April 2022 3 there were 231 bsAbs in clinical trials and a recent industry report 4 predicts the global market revenue for these products will exceed $20 billion by 2028. established Protein A affinity chromatography purification methods for initial product capture.

Pharmaceutical Technology
NOVEMBER 24, 2022
Additionally, the transfer of technology will build and validate capacity for scaling up Good Manufacturing Process (GMP), domestic production of clinical trial products and end-to-end vaccine manufacturing in Africa for regional and global use.

Pharmaceutical Technology
AUGUST 25, 2022
Sonoma Biotherapeutics has signed a lease agreement for setting up an 83,000ft² research and development (R&D) and manufacturing centre for cell therapies. A cGMP-compliant production facility will be housed at the centre for manufacturing cell products at scale.

European Pharmaceutical Review
MARCH 30, 2023
This will increase production capacity four-fold for critical gene therapy and vaccine components. Touchlight – £14 million investment to establish commercial scale manufacture and address the production bottleneck of advanced therapies including mRNA vaccines, gene and cell therapies.

pharmaphorum
DECEMBER 4, 2020
Pfizer slashed its production targets for its COVID-19 vaccine because of a lack of raw materials for its supply chain, according to press reports. A Pfizer spokesperson told Reuters that the “scale-up of the raw material supply chain took longer than expected”. million doses.

European Pharmaceutical Review
JUNE 27, 2022
Alexion , AstraZeneca’s Rare Disease group, will invest €65 million to enhance its production capabilities in Ireland. The investment will help the company scale up its biologics manufacturing capacity and expand R&D efforts over the next 18 months.

Pharmaceutical Technology
APRIL 6, 2023
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has granted ADvantage Therapeutics’ immunotherapy AD04 an Innovation Passport for the treatment of Alzheimer’s disease. A recent wave of monoclonal antibodies, including the FDA-approved Eisai / Biogen ’s Leqembi (lecanemab), is expected to shake up the space.
ISPE
MAY 9, 2023
Product Lifecycle Innovation through Cross-Fertilising Methodological Enablers across Biological Modalities Trudy Patterson Tue, 05/09/2023 - 12:04 iSpeak Blog iSpeak Product Lifecycle Innovation through Cross-Fertilising Methodological Enablers across Biological Modalities Phillip R.

European Pharmaceutical Review
DECEMBER 18, 2023
The Novo Nordisk Foundation has committed up to DKK 1.8 Novo Nordisk Foundation invests in Cellerator, a large-scale cell therapy production site in Denmark NIVI structure NIVI will be made up of two entities, which together will ensure a smooth path between basic R&D up to Phase II clinical trials.

European Pharmaceutical Review
JULY 26, 2023
Cutting-edge equipment and advanced technologies will support the development, manufacturing, and testing of advanced therapy medicinal products (ATMPs). The site will also feature dedicated Class A to D cleanroom areas for bulk drug substances as well as final drug product manufacture.

European Pharmaceutical Review
FEBRUARY 21, 2023
Crucially, January 2023 marked a key moment: the Medicines and Healthcare products Regulatory Agency (MHRA) granted Celadon Pharmaceuticals the first good manufacturing practice (GMP) registration since legalisation in 2018 for high Δ9-tetrahydrocannabinol (THC) cannabis active pharmaceutical ingredient (API). What were the key lessons?
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?

Let's personalize your content