To Improve Retail Pharmacy, Look Beyond Figures and Dig Into Facts
Drug Topics
JANUARY 6, 2023
A local West Virginia mechanic exemplifies the kind of problem-solving that pharmacy executives from major chains should apply to their stores.

Drug Topics
JANUARY 6, 2023
A local West Virginia mechanic exemplifies the kind of problem-solving that pharmacy executives from major chains should apply to their stores.

Pharmacy Times
JANUARY 6, 2023
Lecanemab (Lequembi) approved to treat mild cognitive impairment or mild dementia associated with Alzheimer disease.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

Drug Topics
JANUARY 6, 2023
Your weekly roundup of the latest news from Drug Topics®.
Pharmacy Times
JANUARY 6, 2023
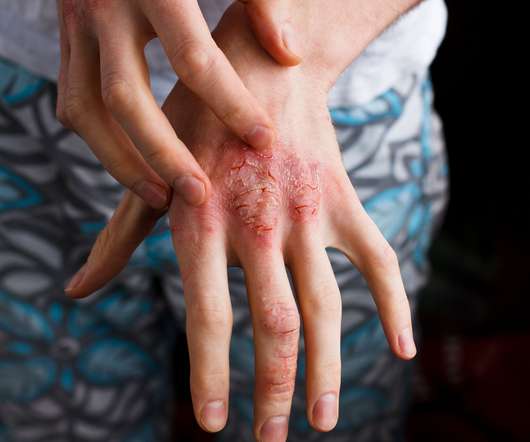
Results show an association between linoleic acid, found in nuts, seeds, and vegetable oils, and increased hypersensitivity in psoriatic lesions.

Speaker: Simran Kaur, Founder & CEO at Tattva Health Inc.
The healthcare landscape is being revolutionized by AI and cutting-edge digital technologies, reshaping how patients receive care and interact with providers. In this webinar led by Simran Kaur, we will explore how AI-driven solutions are enhancing patient communication, improving care quality, and empowering preventive and predictive medicine. You'll also learn how AI is streamlining healthcare processes, helping providers offer more efficient, personalized care and enabling faster, data-driven

Drug Topics
JANUARY 6, 2023
The US Food Administration has agreed to review an application for nirsevimab (Beyfortus; AstraZeneca and Sanofi) to protect infants against RSV.

pharmaphorum
JANUARY 6, 2023
Google and DeepMind have developed an artificial intelligence-powered chatbot tool called Med-PaLM designed to generate “safe and helpful answers” to questions posed by healthcare professionals and patients. The tool is an example of a large language model or LLM, which are designed to understand queries and generate text responses in plain language, drawing from large and complex datasets – in this case, medical research.
Pharmacy Technician Pulse brings together the best content for pharmacy technicians from the widest variety of industry thought leaders.

PharmaVoice
JANUARY 6, 2023
Leaders in the space reveal the topics that are top of mind as the year’s biggest healthcare conference kicks off next week.

Fierce Healthcare
JANUARY 6, 2023
Carbon Health announces another round of layoffs, corporate restructuring amid challenging market conditions. hlandi. Fri, 01/06/2023 - 20:01.
Pharmacy Times
JANUARY 6, 2023
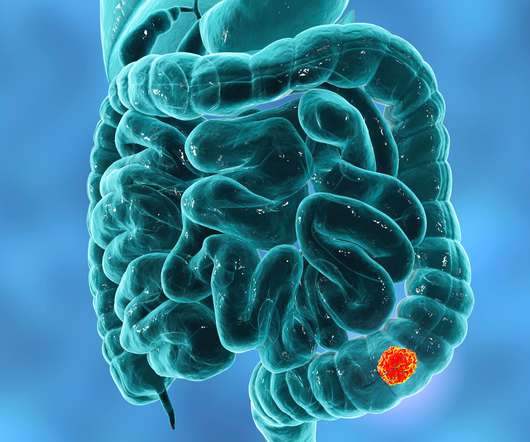
The findings show promising clinical activity and a favorable tolerability profile in the treatment of KRASG12C-mutated advanced colorectal cancer.

Fierce Healthcare
JANUARY 6, 2023
JPM23, Day 1: CVS Health backs $25M investment in Array Behavioral Care. hlandi. Fri, 01/06/2023 - 16:50.

Speaker: Chris Antypas and Josh Halladay
Access to limited distribution drugs and payer contracts are key to pharmacy expansion. But how do you prepare your operations to take the next step? Meaningful data: Collect and share clinical data regarding outcomes, utilization, and more Reporting: Limited distribution models require efficient tracking and reporting systems Workflows: Align workflows with specific pharma and payer contractual requirements For in-depth, expert insights on pharmacy expansion, watch this webinar from Inovalon.
Pharmacy Times
JANUARY 6, 2023
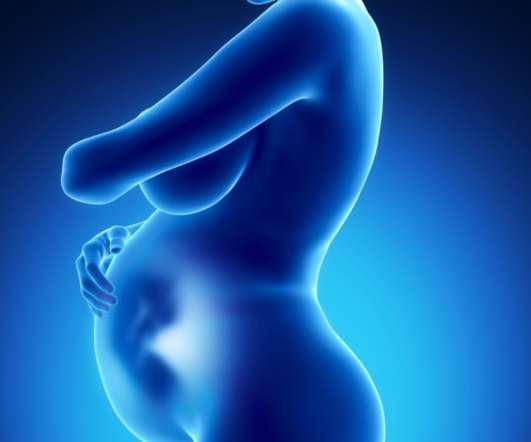
Pharmacists are ideally positioned within communities to identify patients struggling with perinatal depression.
Fierce Healthcare
JANUARY 6, 2023
Industry Voices—Pushing toward a new standard in kidney care. pminemyer. Fri, 01/06/2023 - 11:13.
Pharmacy Times
JANUARY 6, 2023
MenABCWY combines components of 2 vaccines, which help protect against serogroups that cause most cases of the invasive disease.
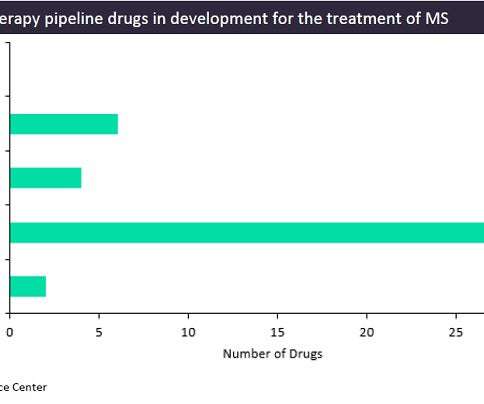
Pharmaceutical Technology
JANUARY 6, 2023
While the treatment options for multiple sclerosis (MS) patients are growing each year with the approval of new agents, all of the currently marketed treatments only slow the disease’s progression and sometimes carry risks of severe side effects, such as liver failure or the development of viral infections. However, new mechanisms of action (MoAs) are in constant development, with some of the more innovative ones utilizing cell-based therapies.

Speaker: Dr. Ben Locwin - Biopharmaceutical Executive & Healthcare Futurist
What will the future hold for clinical research? A recent draft from the FDA provides valuable insight. In "Optimizing the Dosage of Human Prescription Drugs and Biological Products for the Treatment of Oncologic Diseases," the FDA notes that "targeted therapies demonstrate different dose-response relationships compared to cytotoxic chemotherapy, such that doses below the Maximum Tolerated Dose (MTD) may have similar efficacy to the MTD but with fewer toxicities.
Pharmacy Times
JANUARY 6, 2023
Ublituximab-xiiy from TG Therapeutics Inc is the first anti-CD20 monoclonal antibody approved for individuals with RMS that is administered as a 1-hour infusion after the starting dose.

pharmaphorum
JANUARY 6, 2023
From enabling patient choice during clinical trials to strengthening vital partnerships across the quality ecosystem, connected data will become the lifeblood that enables life sciences teams to collaborate efficiently and effectively in 2023. Veeva’s industry experts share their predictions about how breaking down silos across clinical, regulatory, safety, and quality teams will benefit patients.

STAT
JANUARY 6, 2023
Now that the U.S. Food and Drug Administration has granted accelerated approval to the widely anticipated Alzheimer’s drug from Eisai and Biogen, a key question is the extent to which payers — private and public — will cover the treatment. Such decisions are based on myriad factors, starting with the average $26,500 price tag. But there are other considerations, including the quality of the clinical trial data, side effect concerns, the patient population for which the medic

Fierce Healthcare
JANUARY 6, 2023
Researchers crawled search engines and searched the dark web to find out the true extent of healthcare ransomware attacks. aburky. Fri, 01/06/2023 - 13:25.

Advertisement
Are you still using workarounds to manage your daily operations? To achieve peak performance, it's time to explore other options for specialty and infusion pharmacy software. Streamline pharmacy operations and improve clinical performance with automated processing, real-time data exchange, and electronic decision support. Download this helpful infographic to: Drive efficiency and patient adherence from referral receipt to delivery and ongoing care – all with our Pharmacy Cloud.

Drug Store News
JANUARY 6, 2023
Rimmel London’s new launches include an Eye Definer, Brow Definer and two mascaras.

PharmExec
JANUARY 6, 2023
One company pursues a program for a technology they believe is a potential cure for Type 1 diabetes.

European Pharmaceutical Review
JANUARY 6, 2023
The US Food and Drug Administration (FDA) has granted priority review for glofitamab, Roche’s CD20xCD3 T-cell engaging bispecific antibody. The decision for the FDA priority review of glofitamab is for adults with relapsed or refractory (R/R) large B-cell lymphoma (LBCL) after two or more lines of systemic therapy. If approved, the cancer immunotherapy would be the first fixed-duration, off-the-shelf CD20xCD3 T-cell engaging bispecific antibody available for individuals with an aggressive

pharmaphorum
JANUARY 6, 2023
With the US economy teetering on a recession, sales and marketing budgets are expected to take a hit among businesses across sectors. The pharmaceutical industry is no exception. When any organisation looks to slash its budget, the old adage about acquiring new customers costing five times as much as retaining existing customers becomes an alluring data point.

STAT
JANUARY 6, 2023
The pharmaceutical industry has been slow to adopt a business practice that is faster, safer, and more efficient than its current way of operating; that the FDA has been encouraging the industry to implement more widely for years; and that has been used effectively in other industries for decades. I’m talking about continuous manufacturing. Continuous manufacturing produces a drug product from start to finish through a nonstop, integrated assembly.

Fierce Healthcare
JANUARY 6, 2023
Elevance Health rolls out a nutrition tracker in its Sydney member app. pminemyer. Fri, 01/06/2023 - 15:38.
STAT
JANUARY 6, 2023
The Food and Drug Administration on Friday approved a new Alzheimer’s disease treatment that moderately slows cognitive decline in people with early-stage disease. The drug, called Leqembi, was developed by Eisai , the Japanese pharmaceutical company that also developed the first symptomatic treatment for Alzheimer’s 25 years ago.

The Checkup by Singlecare
JANUARY 6, 2023
Mounjaro (tirzepatide) is a brand-name prescription medication that has been approved by the Food and Drug Administration (FDA) to improve blood sugar control in adults with Type 2 diabetes (along with diet and exercise). More recently, Mounjaro has become increasingly popular for weight loss. “In clinical trials Mounjaro has reduced patients’ weights by more than 20%,” explains Suzanne Soliman, Pharm.D., a board-certified pharmacist and founder of Pharmacist Moms Group.

STAT
JANUARY 6, 2023
The Environmental Protection Agency announced on Friday more aggressive air quality standards for particulate matter, pollutants small enough to be inhaled and cause respiratory illness and other disease. The agency’s new rules would help clean the nation’s air and bring it more in line with the past decade of research on the harmful effects of particulate matter.

Drug Store News
JANUARY 6, 2023
The Clinical-Strength Superfoods line consists of capsules that contain herbs, the company said.

STAT
JANUARY 6, 2023
Hired someone new and exciting? Promoted a rising star? Finally solved that hard-to-fill spot? Share the news with us, and we’ll share it with others. That’s right. Send us your changes, and we’ll find a home for them. Don’t be shy. Everyone wants to know who is coming and going. And here is our regular feature in which we highlight a different person each week.

PharmExec
JANUARY 6, 2023
A chat with with Indegene's Timothy Moore, Senior Vice President, Emerging Biotech and Key Accounts and Jack Mycka, Vice President, Enterprise Medical Solutions and Emerging Biotech.

pharmaphorum
JANUARY 6, 2023
pharmaphorum is proud to announce its inaugural “The World Without Disease Summit”: a high quality, two-day event bringing together key global leaders and innovators to explore the potential of disease interception in future health. The event has been conceived in partnership with an established industry leader in this space, Ben Wiegand, Founding Partner at CWWDA (The Connected World Without Disease Accelerator) and former senior executives at UnitedHealth and Janssen.

European Pharmaceutical Review
JANUARY 6, 2023
GSK will exclusively license up to four bi- and multi-specific T cell engaging (TCE) antibodies developed using technology platforms from WuXi Biologics, a contract research, development, and manufacturing organisation (CRDMO). Licensing T cell engaging antibodies. The CRDMO will receive a $40 million upfront payment from GSK and up to $1.46 billion in additional payments for research, development , regulatory and commercial milestones across the four TCE antibodies.
STAT
JANUARY 6, 2023
And so, another working week will soon draw to a close. Not a moment too soon, yes? This is, you may recall, our treasured signal to daydream about weekend plans. Our agenda is still being assembled, but we do know that we will promenade extensively with the official mascot. We also hope to catch up on our reading, since our stack of overdue library books is disheartening.
Let's personalize your content