Driving QbD as a quality standard in drug development
European Pharmaceutical Review
JANUARY 2, 2024
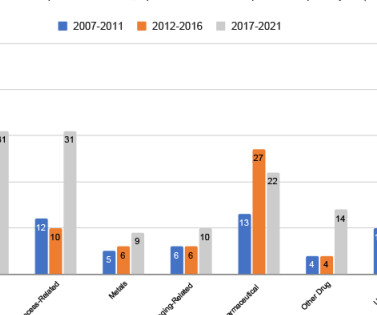
A paper published in the European Journal of Pharmaceutics and Biopharmaceutics has reviewed the current application status of Quality by Design (QbD) within the framework of the ICH guidelines (ICH Q8(R2) – Q14 and ICH Q2(R2)). The team examined the European Public Assessment Report (EPAR) for the QbD approach.




























Let's personalize your content