Considerations for safety data migration methods
European Pharmaceutical Review
SEPTEMBER 26, 2023
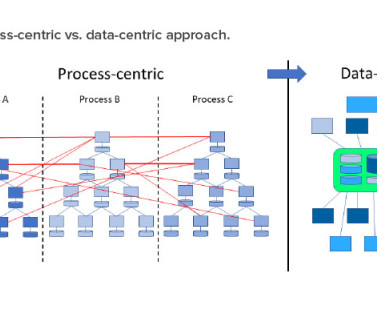
During migration, the case processing team will manually enter the data using the outputs obtained from the source database, following the workflow configuration, to ensure accuracy within the target database.









Let's personalize your content