Considerations for a Decentralized Manufacturing Paradigm
ISPE
SEPTEMBER 11, 2023
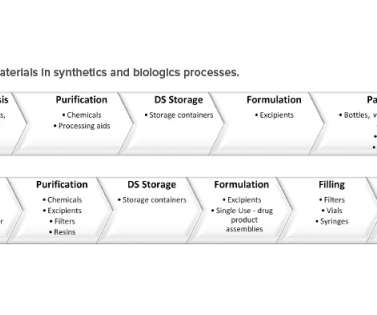
Examples include platforms as small as autonomous units placed within a facility and mobile production trailers for compounding to platforms as large as a prefabricated, self-contained GMP facility. FDA Official Warns Manufacturers of Common Problems Found in Aseptic Operations.” September 2022. 7 March 2023. Regulatory Focus.










Let's personalize your content