Considerations for a Decentralized Manufacturing Paradigm
ISPE
SEPTEMBER 11, 2023
This article presents relevant insights on the current regulatory and technical landscape for decentralized manufacturing, with select examples of current applications, and discusses perspectives on evolving and adapting the current regulations to meet future capabilities. Product Focus: Advancing Aseptic Processing.” 5 October 2022.




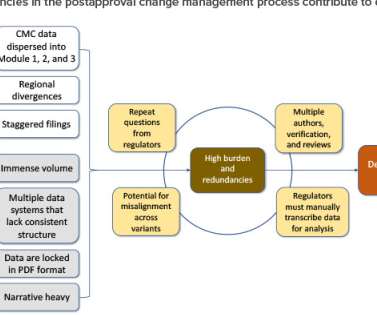
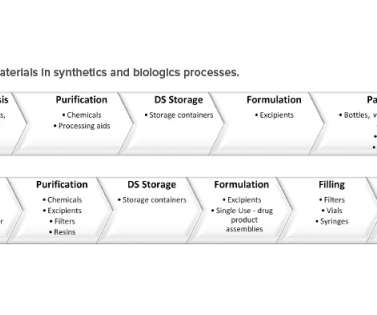
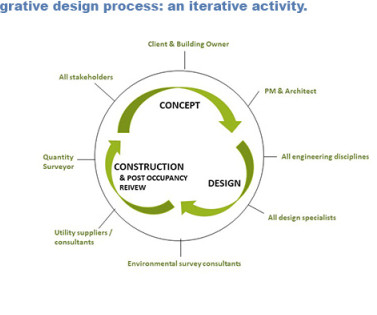







Let's personalize your content