CMC Requirements for New Drug Registration in Latin America
ISPE
MAY 9, 2023
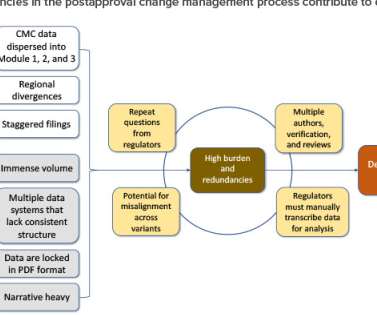
Improvements have been observed by various regulatory agencies in Latin America for the acceptance and implementation of international standards—for instance, the ICH Common Technical Document (CTD) format. This article offers an overview of the CMC requirements for the small molecules product registration process in Latin America.








Let's personalize your content