Delivery systems for biologics
European Pharmaceutical Review
MARCH 4, 2024
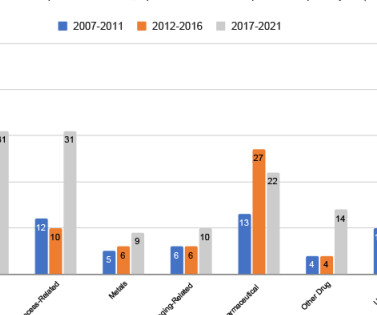
These biopharmaceuticals encompass a range of products including vaccines, recombinant therapeutic proteins, tissues, cells and gene therapies – all sourced from living organisms. Indeed, in 2022, biologics constituted 40 percent of all US Food and Drug Administration (FDA) approved drugs, projecting a compound annual growth rate (CAGR) of 9.5









Let's personalize your content